
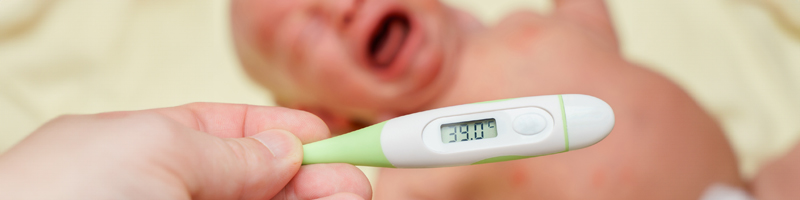
Emergency clinicians frequently provide care to febrile infants aged ≤60 days in the emergency department. In these very young infants, fever may be the only presenting sign of invasive bacterial infection and, if untreated, invasive bacterial infection can lead to severe outcomes. This issue reviews newer risk-stratification tools and the 2021 American Academy of Pediatrics clinical practice guideline to provide recommendations for the evaluation and management of febrile young infants. The most recent literature assessing the risk of concomitant invasive bacterial infection with urinary tract infections or positive viral testing is also reviewed.
How would you manage these patients? Subscribe for evidence-based best practices and to discover the outcomes.

Subscribe to access the complete flowchart to guide your clinical decision making.

Subscribe for full access to all Tables.
Buy this issue and
Following are the most informative references cited in this paper, as determined by the authors.
1. * Pantell RH, Roberts KB, Adams WG, et al. Evaluation and management of well-appearing febrile infants 8 to 60 days old. Pediatrics. 2021;148(2):e2021052228. (Clinical practice guideline) DOI: 10.1542/peds.2021-052228
10. * Gomez B, Mintegi S, Bressan S, et al. Validation of the “Step-by-Step” approach in the management of young febrile infants. Pediatrics. 2016;138(2):e20154381. (Prospective; 2185 patients) DOI: 10.1542/peds.2015-4381
11. * Kuppermann N, Dayan PS, Levine DA, et al. A clinical prediction rule to identify febrile infants 60 days and younger at low risk for serious bacterial infections. JAMA Pediatr. 2019;173(4):342-351. (Prospective; 1821 patients) DOI: 10.1001/jamapediatrics.2018.5501
41. * Aronson PL, Shabanova V, Shapiro ED, et al. A prediction model to identify febrile infants ≤60 days at low risk of invasive bacterial infection. Pediatrics. 2019;144(1):e20183604. (Case-control; 543 patients) DOI: 10.1542/peds.2018-3604
43. * Milcent K, Faesch S, Gras-Le Guen C, et al. Use of procalcitonin assays to predict serious bacterial infection in young febrile infants. JAMA Pediatr. 2016;170(1):62-69. (Prospective; 2047 patients) DOI: 10.1001/jamapediatrics.2015.3210
44. * Kimberlin DW, Lin CY, Jacobs RF, et al. Natural history of neonatal herpes simplex virus infections in the acyclovir era. Pediatrics. 2001;108(2):223-229. (Prospective; 186 patients) DOI: 10.1542/peds.108.2.223
48. * Burstein B, Alathari N, Papenburg J. Guideline-based risk stratification for febrile young infants without procalcitonin measurement. Pediatrics. 2022;149(6):e2021056028. (Prospective; 957 patients) DOI: 10.1542/peds.2021-056028
64. * Levine DA, Platt SL, Dayan PS, et al. Risk of serious bacterial infection in young febrile infants with respiratory syncytial virus infections. Pediatrics. 2004;113(6):1728-1734. (Prospective; 1248 patients) DOI: 10.1542/peds.113.6.1728
91. * Tzimenatos L, Mahajan P, Dayan PS, et al. Accuracy of the urinalysis for urinary tract infections in febrile infants 60 days and younger. Pediatrics. 2018;141(2). (Prospective; 4147 patients) DOI: 10.1542/peds.2017-3068
96. * Burstein B, Sabhaney V, Bone JN, et al. Prevalence of bacterial meningitis among febrile infants aged 29-60 days with positive urinalysis results: a systematic review and meta-analysis. JAMA Netw Open. 2021;4(5):e214544. (Systematic review and meta-analysis) DOI: 10.1001/jamanetworkopen.2021.4544
98. * Mahajan P, VanBuren JM, Tzimenatos L, et al. Serious bacterial infections in young febrile infants with positive urinalysis results. Pediatrics. 2022;150(4):e2021055633. (Prospective; 7180 patients) DOI: 10.1542/peds.2021-055633
103. *Aronson PL, Louie JP, Kerns E, et al. Prevalence of urinary tract infection, bacteremia, and meningitis among febrile infants aged 8 to 60 days with SARS-CoV-2. JAMA Netw Open. 2023;6(5):e2313354. (Cross-sectional; 14,402 patients) DOI: 10.1001/jamanetworkopen.2023.13354
111. Gutman CK, Aronson PL, Singh NV, et al. Race, ethnicity, language, and the treatment of low-risk febrile infants. JAMA Pediatr. 2023. DOI: 10.1001/jamapediatrics.2023.4890 (Retrospective; 4042 patients)
121. *Brower LH, Wilson PM, Murtagh Kurowski E, et al. Using quality improvement to implement a standardized approach to neonatal herpes simplex virus. Pediatrics. 2019;144(2):e20180262. (Quality improvement) DOI: 10.1542/peds.2018-0262
Subscribe to get the full list of 127 references and see how the authors distilled all of the evidence into a concise, clinically relevant, practical resource.
Keywords: fever, febrile young infant, febrile infant, fever in infants, pediatric fever, neonatal fever, serious bacterial infection, SBI, invasive bacterial infection, IBI, urinary tract infection, UTI, neonatal herpes simplex virus, herpes simplex virus, Febrile Young Infant Research Collaborative, infant younger than 60 days, bacteremia, bacterial meningitis, Escherichia coli, E coli, Staphylococcus aureus, Listeria monocytogenes, enterovirus, parechovirus, SARS-CoV-2, urinalysis, urine culture, blood culture, CSF culture, CSF pleocytosis, risk stratification, biomarkers, procalcitonin, PCT, temperature, absolute neutrophil count, ANC, C-reactive protein, CRP, antibiotic therapy
Lauren Palladino, MD, MSHP; Christopher Woll, MD, FAAP; Paul L. Aronson, MD, MHS
Jeffrey R. Avner, MD, FAAP; Kate Dorney, MD, MHPEd
February 1, 2024
February 1, 2027 CME Information
4 AMA PRA Category 1 Credits™, 4 ACEP Category I Credits, 4 AAP Prescribed Credits, 4 AOA Category 2-B Credits. Specialty CME Credits: Included as part of the 4 credits, this CME activity is eligible for 1 Pharmacology and 1 Infectious Disease CME credit, subject to your state and institutional approval.