
|
|
Emergency physicians (EPs) tend to be action oriented with a penchant for adventure and the outdoors. Consequently, emergency physicians are often actively engaged in high altitude activities and thus positioned for providing care to patients with altitude related illness. Because of the unique skill set, EPs are frequently sought out to be the physicians for trips into the austere high altitude environment and are also asked to provide pre trip guidance. Proper preplanning and monitoring can literally make the difference between life and death. In addition to recreational activities, the reality of the global war against terrorism has brought altitude illness into the mainstream of many military emergency physicians in that combat in Central Asia is often occurring at elevations of 3000 to 4000 meters (m) and higher. 1 This issue of Emergency Medicine Practice provides a comprehensive overview of the spectrum of altitude illness with a focus on prevention strategies and life saving interventions in the eventuality for severe disease.
| ADH | : | Antidiuretic Hormone |
| AMS | : | Acute Mountain Sickness |
| CAD | : | Coronary Artery Disease |
| CNS | : | Central Nervous System |
| CXR | : | Chest X-ray |
| DKA | : | DKA: |
| ESQ-IV | : | Environmental Systems Questionnaire, fourth edition |
| HACE | : | High Altitude Cerebral Edema |
| HAP/B | : | High Altitude Pharyngitis and Bronchitis |
| HAPE | : | High Altitude Pulmonary Edema |
| NSAIDs | : | Non Steroidal Anti Inflammatory Drugs |
| PFO | : | Patent Foramen Ovale |
| PRK | : | Photorefractive Radial Keratectomy |
| LASIK | : | Laser Assisted In Situ Keratomeliusis |
| USARIEM | : | US Army Institute of Environmental Medicine |
A Medline search was performed using the terms:altitude illness, AMS, HAPE, HACE, acetazolamide, altitude sickness, altitude, gingko and altitude, sildenafil and altitude, dexamethasone and altitude, and nifedipine and altitude. References were also obtained from review articles published in the last six years. The Wilderness Medical Societies' resources and the online altitude medicine resourceat www.hypoxia.net were also utilized. Additionally, abstracts from The World Congresses on MountainMedicine and High Altitude Physiology from 2000 to 2006 were reviewed. Neither the Cochrane Database, ACEP Clinical Policies, nor the National Guideline Clearinghouse had guidelines on altitude illness. The only specific evidence-based review found was the Canadian Committee to Advise on Tropical Medicine and Travel (CATMAT) advisory.There have been several excellent reviews on altitude medicine published in the last five years.3-5 Most of these reviews were based on similar original source material; thus, the original sources were utilized where appropriate. Interestingly, there is almost a complete lack of controlled studies on the most effective emergency department therapies for altitude syndromes. While there are well done, double blind, placebo controlled studies on AMS prophylaxis, and some on treatment of AMS and HAPE in the field, treatment of altitude illness in the ED is almost exclusively based on theory, field treatments, and anecdote.
Altitude is typically broken into three levels withgeneral physiologic changes at each level.
There are four large categories of altitude induced illness:
In a 1993 survey involving 3158 respondents, Honigman et al reported that 25% of visitors to an elevation of 2000 to 3200 m developed AMS on arrival. (Table 1 ) 6 The following risk factors for developing AMS were identified:
A 2002 study of mountaineers by Schneider et al did not validate the above risk factors. However, it looked at a select group; i.e., fit mountaineers, many of whom had prior altitude experience; plus, there was a election bias, as only those who had made it

to 4559 m were included.7 The study by Honigan et al is here fore considered a better representation of individuals at risk for altitude illness. Physical fitness level has not been shown to influence the incidence of AMS.8 9
In a 1976 study of 278 Himalyan trekkers at 4243 m, the incidence of AMS was found to be 53% in unacclimatized individuals.10 A 1986 study of trekkers at 5400 m had a similar incidence of 49%. Since these two studies were performed, knowledge of altitude illness and its prevention increased; thus, a subsequent tudy in 2001 reported a decreased incidence of 28%.11 An incidence of 29% was also reported in a 2004 study by Gaillard et al who studied 500 trekkers hiking to 5400 m. The decreased AMS incidence in the 2004 study was probably also due to a slower rate of ascent compared to the 1986 study and increased use of elf-medication for AMS, from 17 to 56%.12
Rapid ascent is a risk factor for AMS. (Figure 1)In one study, 14% of racers in an expedition race who ascended rapidly from 3000 to 4500 m developed AMS which required medical treatment (this did not include those with AMS who did not request formal medical treatment).13 In another study involving rapid ascent in the Alps, the incidence of AMS was 9% at 2850 m, 13% at 3050 m, 34% at 3650 m, and 53% at 4559 m.14 In a case series of 459 climbers in Iran, a rapid ascent from 2900 to 4200 m over four hours resulted in a 61% incidence of AMS.15 A convenience sample of 228 pilgrim trekkers to 4300 m in Nepal found a 68% incidence of AMS, similar to that found on a Mount Rainier ascent.16

This incidence of AMS in children at altitude is also substantial; 28% of children who ascended to 2835 m from 1600 m developed AMS, as determined by a questionnaire of 558 children age nine to 14.17
HAPE represents the severe pulmonary syndrome due to progressive AMS. It is responsible for the greatest number of high altitude fatalities (up to 20 annually18), with a mortality approaching 50% if descent is not undertaken in a timely manner. 19 HAPE occurs two to five days after arrival at a new elevation and is very rare after one week of acclimatization.20 This timing data comes from a European series of 52 patients admitted with HAPE. All had echocardiography, a complete history and exam, and blood ultures performed to rule out other causes of the symptoms.21 All were treated with nasal oxygen, oral nicardipine, and bedrest at a post descent elevation of 500 m. All recovered and were discharged after a mean of four days (+/- 2). In a retrospective study of 47 cases from Vail, Colorado, the mean onset of symptoms was 2.5 0.8 days after arrival at altitude. In all cases, HAPE may come on and progress either insidiously or rapidly over a few hours. Symptoms may develop and worsen at night.22
The incidence of HAPE varies with the altitude achieved and the rate of ascent. Climbers in the Alps who ascend to 4559 m over two to four days have a reported incidence of less than 0.2%;23 however, if the same altitude is reached in 22 hours, the incidence increases to 10%.24 In a report of soldiers airlifted rapidly to 5500 m, the incidence of HAPE has been reported to be 15.5%.25 A study from Vail, Colorado 2 reported an incidence of 0.01 to 0.1% amongst skiers at the lower elevations of 2500 to 3000 m.26
The incidence of subclinical pulmonary edema may also be significant. In an observational study carried out at 4559 m in the Alps, 15% of climbers were found to have rales or interstitial edema on chest xray; 92% of this group also had decreased pulmonary closing volumes, confirming that these subjects did have HAPE. Seventy-four percent of all other asymptomatic climbers also had decreased closing volumes, suggesting up to three-quarters of all climbers may have some degree of pulmonary overload, though this is of questionable clinical significance.27,28
Individuals found to be susceptible for HAPE have a significant risk of recurrence, with a reported incidence of 66% on subsequent altitude exposures.29 Susceptibility to HAPE may be predictable by testing an individual's pulmonary pressure response to sea level hypoxic testing or to exercise testing; however the sensitivity (77 to 94%) and specificity (76 to 93%) are not well established.30 The young appear to be at the highest risk of HAPE.31,32
Conditions which either increase the pulmonary arterial pressure or affect the blood/lung barrier can increase the risk of developing HAPE.33-35 HAPE incidence is likely higher in children just before or after an upper espiratory infection. This has also been seen in animal studies.36,37 Vigorous exercise, because it elevates pulmonary artery pressure, may increase HAPE risk in those susceptible and, therefore, should be avoided for the first few days at altitude.38 There has also been an association of patent foramen ovale (PFO) and HAPE, an observational study of 25 HAPE susceptible individuals found that the incidence of PFO was three times higher in HAPE susceptible persons versus those not susceptible.
HACE was first described in 1913 by Ravenhill as "Nervous Punta," a central nervous system (CNS) form of mountain sickness.40 It represents the neurological syndrome of severe AMS and is the least common form of altitude illness. A case series of 1925 soldiers rapidly transported 3350 to 5000 m reported an incidence of .2%.71 In the Alps, the HACE incidence is reported as 1:588 at 4559 m and 1:4000 at 2795 to 3050 m.41 A case series in 1976 found a similar incidence of 1.8% out of 278 trekkers at 4243 m.53 The incidence of climbers on Denali is reported at 2 to 3%.42 A convenience sample from Nepal reported the highest incidence, with a HACE rate of 31%; this is the only study to show a higher incidence of HACE than HAPE which is a finding that the authors could not explain. The study was a convenience sample of 228 out of 5000 subjects, and the authors concluded that the high altitude pilgrims sampled may, for some reason, represent a highly susceptible group. HACE typically occurs three to five days after arrival at elevations higher than 3000 m, with a mean altitude onset of 4730 m. The lowest altitude at which HACE has been reported is 2100 m.43 this same review, the first done on HACE since the 1970s, gives 24 to 36 hours as the typical time of progression rom AMS to HACE. Coma may occur as soon as 24 hours after ataxia is noted.4
Hypobaric hypoxia is the term used to describe thehypoxia of altitude which is due to the exponential decrease in barometric pressure with subsequent decrease in the partial pressure of oxygen. (Figure 2) The oxygen concentration of the atmosphere remains nearly constant at 21% up to an elevation of 8800 m. It is the decreased partial pressure with subsequent decrease in the partial pressure of oxygen rather than a decreased concentration which causes the hypoxia of altitude. Acclimatization is the process by which the body physiologically adapts to the hypobaric hypoxia of altitude. This is a complex process which begins at elevations above 1500 m,(hence why thedefinition of altitude begins at 1500 m of elevation).
The first response of an individual to altitude is increased ventilation in an effort to reduce the hypoxia. This response is mediated by the carotid body chemo-receptors and results in a blowing off of alveolar CO2 which then allows the retention of alveolar oxygen.44 Hyperventilation results in a respiratory alkalosis which stimulates the respiratory center in the medulla to inhibit the ventilatory rate. There is then a somewhat compensatory metabolic acidosis which results from the increased renal excretion of bicarbonate, allowing for continued hyperventilation. There physiologic processes, such as increasing red cell mass, changes in delivery of oxygen to tissues, and uptake of oxygen in the lungs, are also involved in acclimatization. The process of acclimatization requires time for these physiologic processes to occur. Given a gradual increase in elevation, the body can adapt effectively; failure to provide adequate time for acclimatization increases the risk for illness.
In order to promote acclimatization, it is generally recommended that after 3000 m, the sleeping elevation be advanced only by 300 m a day with a rest day every third day.45 A study by the US Army Institute of Environmental Medicine (USARIEM) showed that returning to sea level results in a gradual loss of the acclimatization.46 However, the researchers also reported that, following two to threeweeks of acclimatization at 4300 m, a return to sea level for eight days did not result in a loss of acclimatizedexercise ability. Intermittent altitude exposure has also been shown to result in acclimatization. In one experimental study using six subjects subjected to low intensity exercise, intermittent altitude exposure in a chamber for three to five hours a day for 17 days reduced AMS on subsequent altitude exposure. 47 In another USARIEM study it was shown that exercise was not required for this effect as intermittent exposure to altitude for four hours a day five days a week for three weeks at 4300 m effectively reduced AMS severity.48 Acclimatization to altitude does require exposure to hypobaric hypoxia; i.e., breathing normobaric hypoxic gas at sea level does not decrease the incidence of AMS.49
While failure to acclimatize results in AMS, the pathophysiology is complex, involving generalized fluid retention, hypoventilation, and CNS changes. Fluid retention is associated with AMS, generally developing within three hours and resulting in a rise of ADH. Individuals who do not develop AMS do not retain fluid on altitude exposure and have been found to have a rapid drop in ADH.50 The actual hydration status of an individual does not appear to affect the development of AMS. 51
Failure to hyperventilate on altitude exposure is another factor associated with AMS. Individuals who develop AMS appear to have a relativehypoventilation as compared to those who do not develop AMS, and are relatively hypoxic compared to those who are not sick.52,53
Hypoxemia-related CNS swelling resulting in increased cerebral blood volumes has also been implicated in AMS.54 In cases of moderate to severe AMS, an MRI study of seven individuals found four of the subjects to have an increase T2 signal in the brain white matter (these subjects did not have clinically definable HACE as they had no ataxia or alteration in mental status).55 The presence of brain swelling alone, however, does not correlate with AMS; a 1999 study by Muza et al showed that, in seven ubjects who ascended to 5000 m in a chamberfor eight hours, all had increased brain volume on MRI regardless of whether they had AMS or not.56 It is unclear why all individuals develop brain swelling at altitude but only some individuals develop AMS. One hypothesis is that AMS is related to craniospacial capacitance to accommodate swelling; i.e., those with limited capacitance who cannot accommodate the cerebral swelling of altitude develop AMS, while those who can accommodate it do not.57 This hypothesis also might explain why the elderly, with their decreased brain volumes, have a lower incidence of AMS.58
HAPE susceptible individuals show an exaggerated pulmonary artery pressure elevation on exposure to hypoxia and to exercise in both normoxic and hypoxic environments.59 This exaggerated pulmonary response, which initially results in non-homogeneous vasoconstriction and regional overperfusion, is a hallmark of those susceptible to HAPE, but this factor alone is not sufficient to cause HAPE.60 Cardiaccatheterization of HAPE victims shows a normal wedge pressure but a markedly elevated pulmonary artery pressure of 60 mm on average.61 Broncheoalveolar lavage of patients with HAPE shows high protein concentrations, aracadonic acid metabolites, and complement activation products. The edema to serum protein ratio is 0.7:1, demonstrating that HAPE represents a break down in the alveolar/endothelial barrier. 62 These findings show that HAPE is a result of a change in pulmonary vascular endothelial permeability and increased pulmonary arterial pressures, and not a problem of fluid overload. Treatments of cardiac etiology traditionally used for pulmonary edema are therefore relatively ineffective for treating HAPE.
Other changes in vascular permeability are also required for HAPE to develop. Recent research points to changes in sodium-dependent absorption of liquid from the airways which may be defective in patients who are susceptible to high-altitude pulmonary edema,63 as well as nitric oxide production deficiencies in those susceptible to HAPE.64,65
Increased intra-cranial pressure may also play a role in the development of HAPE. Pulmonary edema has been shown to be a pathophysiological consequence of increased inter cranial pressure.66-68 It has also been shown that those who are HAPE susceptible have an exaggerated sympathetic response (a two to three fold increase in sympathetic discharge compared to controls) to the hypoxia of altitude. 69 Those susceptible to HAPE also have an increase of pulmonary artery pressure when exposed to the cold as compared to those who are not HAPE susceptible.70
The onset of AMS is rapid on exposure to altitude, with 65% of patients having an onset of symptoms within 12 hours and another 34% within 36 hours; only 1% of individuals have an onset after 36 hours.6 Typically, mild AMS is self limited if further ascent is not undertaken. The best longitudinal study of AMS duration was in the 1960s involving Indian Soldiers flown rapidly to 3300 to 5500 m. Sixty percent of the soldiers developed AMS, with symptoms lasting two to five days; 40% had persistent symptoms at one week which decreased to 13% by one month.71 Most subsequent reviews give duration of two to five days for mild AMS.72,73
Clinically, AMS presents with headache (typically frontal, bitemporal and worsens at night and with exertion),74 GI symptoms (emesis, nausea, and anorexia), insomnia, fatigue/weakness, and dizziness/ lightheadedness. 75,76 Several standardized clinical scoring systems have been utilized to determine if a subjects' symptoms represent AMS. Historically, the standard scoring system was the USARIEM developed Environmental Systems Questionnaire, currently in its fourth edition (ESQ-IV). A simpler method is the Lake Louise acute mountain sickness questionnaire. (Table 2) Both are effective tools for determining AMS,77 though the ESQ-IV does not require headache as a minimum and is the more conservative of the two.78 In essence, the Lake Louis consensus definition of altitude illness sets the current standard definition for AMS.

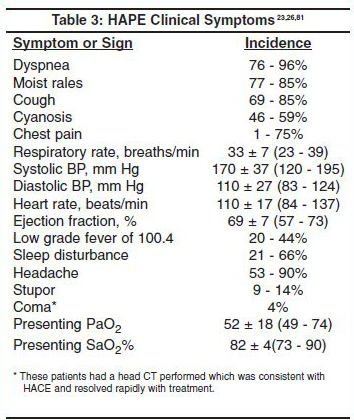
The clinical symptoms of HAPE are similar to pulmonary edema of non altitude causes, despite the completely different etiologies. These are tachypnea, dyspnea, cough, cyanosis, moist rales (unilateral or bilateral), decreased PaO2 and arterial oxygen saturation (with low or normal PaCO2), and pulmonary infiltrates on x-ray (unilateral or bilateral.)79 The first symptoms of HAPE are decreased exercise tolerance and prolonged xercise recovery time.80 (Table 3)

As with AMS, the case definition of HAPE was codified by the Lake Louise Consensus Committee; an individual is defined as having HAPE if they meet the criteria in Table 4. HAPE severity is classified in four grades based on signs and symptoms. (Table 5) 82


AMS is present before HACE as HACE progresses from AMS. Drowsiness and subtle psychological changes are the first clinical findings to appear. These symptoms then progress to more significant altered mental status which ranges from behavioral changes, including irrational or bizarre behavior, to progressive lethargy and coma. Ataxia is a prominent clinical finding and it has been stated that "if a patient seems mildly drunk at altitude, they have cerebral edema."84 Headache is prominent and papilledema may be seen. (Table 6) HAPE often coexists with HACE, with 11 of the 13 cases from one study suffering from both. The Lake Louise Consensus has also set criteria for the diagnosis of HACE. (Table 7)


The differential diagnosis of AMS is broad which can lead to a delay in diagnosis and proper management. The most common considerations are influenza like illness, viral syndromes, exhaustion, vascular headache, dehydration, alcohol hangover, carbon monoxide toxicity, substance abuse, and hypothermia. 3,4
The differential diagnosis of HAPE includes pulmonary edema of any cause (cardiogenic, neurogenic, toxic, immersion, etc) as well as congestive heart failure, myocardial infarction or cardiac ischemia, pulmonary embolism, reactive airway disease, COPD, pneumonia, and bronchitis.3-4
The differential diagnosis of HACE includes the full spectrum of diseases that cause altered mental status, including carbon monoxide poisoning, stroke, metabolic disorders, CNS infections, CNS lesions, and intoxications.3
The vast majority of AMS cases will be encountered in the prehospital environment, and proper recognition and treatment will have significant impact on outcome. Mild to moderate AMS is typically self limited, provided that one does not continue to ascend in elevation. If AMS develops, individuals should not ascend further; in particular, it is recommended that the sleeping elevation not be increased until symptoms improve. Decent is very effective but not mandatory. If undertaken, a descent of 500 m is recommended. For mild AMS, it is likely that only symptomatic treatment will be required, though medications specific for AMS may also be utilized. Symptomatic treatment includes treatment of headache, nausea, and vomiting. Headache treatment includes aspirin, acetaminophen, or NSAIDs.86,87 Acetaminophen 1000 mg has been shown to be as effective as 400 mg of ibuprofen for symptomatic relief of high altitude headache.88 Nausea and emesis have typically been treated with prochlorperazine, 10 mg intravenously or 25 mg oraly every six hours, or promethazine, 25 mg by rectal suppository or intramuscularly every six hours.89 Prochlorperazine is also a respiratory stimulant as opposed to most other antiemetics which may theoretically have a negative effect on ventilation.
Acetazolamide has been shown to be effective for treating AMS and also aids acclimatization by inducing a metabolic acidosis and compensatory respiratory alkalosis; in effect, inducing the normal acclimatization process in reverse. In a well done study, Grissom showed that, in individuals with AMS, acetazolamide 250 mg at zero and eight hours significantly improved symptoms, alveolar-arterial gradient, and oxygenation. 90 Acetazolamide, 250 mg or 500 mg orally twice a day, is now an accepted treatment dose; it is the only drug with an FDA indication for altitude illness.
Acetazolamide was found in one small study of 13 subjects to be even more effective for AMS treatment when combined with dexamethasone.91 Dexamethasone has also been shown to effectively treat AMS at a dose of 4 mg PO qid.92
For moderate to severe AMS, consensus prehospital treatment recommendations are to add low flow oxygen as available (Class I recommendation, see back page for definitions), hyperbaric therapy as available (Class I), and to consider adding dexamethasone to acetazolamide therapy (Class II), as well as descending 500 m.
HAPE untreated has a high mortality; therefore, proper prehospital care is critical. Supplemental oxygen to bring the oxygen saturation above 90% is a mainstay of treatment. Immediate descent of at least 1000 m is mandatory, if at all possible. Exposure to cold and exertion should be minimized during descent as much as possible as both raise pulmonary artery pressure.93 If descent is not possible, portable hyperbaric chambers, such as the Gamow Bag, have been successfully used to treat HAPE until descent could be accomplished.94,95 Pharmacological therapy may be added to oxygen if available; see "Treatment" section. There is no literature support for use of pharmacologic therapy solely as an alternative to descent for HAPE treatment. Nifedipine is the most often recommended agent. Salmeterol 125 ug bid (or possibly albuterol) has literature support for use, though only salmeterol has thus far been studied.157 Dexamethasone use can be considered, particularly if any HACE or severe AMS coexists.159
For HACE in the prehospital setting, all reviews recommend oxygen, descent, and dexamethasone 8 to 10 mg by any available route (IV, IM, PO), followed by 4 mg every six hours.3,43 Some reviews also suggests adding acetazolamide for additional benefit, though no literature is available to support this. If descent is not an immediate option, then hyperbaric therapy can be an effective temporizing measure. 96
Gather the following historical information from someone with altitude illness: the altitude attained, time spent in acclimatization, any past history of altitude illness and, particularly, the altitude and time frame of onset in any past episodes. A history of a particular altitude illness at a particular altitude in the past is likely to be repeated.
AMS is a symptomatic historical diagnosis; there are no specific findings on physical exam for AMS.97 However, vital signs and physical exam are useful in diagnosing HAPE and HACE, as well as in ruling out other causes of symptoms.
Vital signs are of great utility in the evaluation of HAPE and HACE; tachycardia is typically present in HAPE. Fever is only present in HAPE, not with AMS or HACE.4,98 Pulse oximetry is useful for HAPE which will usually have oxygen saturations lower than expected for a given altitude.
Focus the physical exam predominantly on the lung exam, cardiac exam, and neurological exam. On lung examination, those with HAPE will have rales present; this, however, is not pathognomonic as a study by Hackett found a 23% incidence of rales in Himalayan trekkers, 40% of whom had neither AMS nor HAPE.99 Peripheral and facial edema may be seen in association with AMS or as an isolated benign finding; these findings do not indicate presence of the severe edemas associated with HAPE and HACE.
The cardiac exam is useful in differentiating HAPE from cardiogenic pulmonary edema as an S3 will not be heard in HAPE.100 The neurological exam, specifically assessment of mental status and ataxia, is fundamental to diagnosing HACE. Papilledema is suggestive of an elevated ICP and, thus, also supports a diagnosis of HACE; however, it takes up to 24 hours of elevated ICP before papilledema develops.
There are no pathognomonic laboratory findings in AMS, HACE, or HAPE. In HAPE, arterial saturation is often low and generally correlates with the severity of disease. The white blood cell count may be either normal or elevated with a left shift. 101,102
Urinalysis often reveals proteinuria in AMS, although this finding is of no prognostic significance. 103,104 In HACE, the lumbar puncture opening pressure is generally elevated (i.e., 44 to 220 mm H2O), but cerebrospinal fluid analysis is normal.4
There are no specific radiographic findings associated with AMS. HAPE almost always has radiographic abnormalities present on x-ray, though the clinical symptoms may not always mesh with the degree of radiographic lung involvement.
Early in HAPE, the CT and plain radiographic appearance of the lungs is one of patchy and peripheral edema. As HAPE becomes more advanced, it produces a more homogeneous and diffuse radiographic appearance.105 In one of the early studies on HAPE, Hultgren developed staging criteria to define the radiological severity of HAPE. (Table 8) These radiographic findings correlate with clinical severity. The "bat wing" appearance of cardiogenic pulmonary edema is unusual in HAPE.4 The radiological changes consistent with HAPE are usually present by 18 hours after rapid transport to high altitude, and the findings resolve within 24 to 72 hours after therapy or descent.

On MRI, HACE is seen as white matter edema (increased T2 signal), particularly in the corpus callosum. 106,107 This is consistent with a vasogenic edema as the etiology of HACE. CT or MRI may be confirmatory of the diagnosis of HACE, but these studies are most useful in excluding other etiologies of altered mental status. Because of the high incidence (up to 80%) of HAPE with HACE, a chest x-ray in recommended in patients presenting with HACE.
Generally, AMS has improved by the time an individual arrives in the ED in that they have already descended. Table 9 provides a summary of recommendations that are commonly found in the literature.

AMS is very common, as demonstrated by its high incidence in the many series already referenced. Fortunately, there are a multitude of methods of prophylaxis for AMS and treatment once it occurs. Just as there are many interventions that have been shown effective for AMS, there are also a number that have been shown ineffective (Table 10) When all is said and done, there is no debate in the literature that a slow ascent provides the best protection against AMS.

Theoretically, by changing the carbohydrate/fat/protein content of a diet to increase the carbohydrate content, one should be able to reduce the amount of oxygen required to generate metabolic energy in an individual. Several studies have shown that a high carbohydrate diet will improve oxygen saturation; however, only one small study from the 1970s showedan improvement in AMS incidence.121,122 This has not been born out in more recent studies, however, as a high carbohydrate diet (68% vs. 45% for normal diet) for four days does not decrease the incidence of AMS in those exposed to normobaric hypoxia.123Likewise, in a study in 1999, a high carbohydrate diet improved oxygenation but did not effect the incidence of AMS.124 Therefore, thepreponderance of the literature does not show a decrease in AMS incidence through the use of a high carbohydrate diet.
At the present time, acetazolamide is the drug of choice for AMS prevention and is the only FDA approved medication for this purpose.125,126It is a carbonic anhydrase inhibitor that causes a metabolic acidosis through renal bicarbonate diruesis. As a result, there is increased entilation and oxygenation, which is particularly helpful during sleep when hypoventilation can be pronounced. A secondary benefit of acetazolamide is a decrease in CSF volume and pressure. It first became an accepted agent in the 1960s when a randomized, double blind study involving 43 soldiers transported passively to 3900 m showed a reduction in AMS by 50% in the treated group.127 Subsequent studies confirmed this finding.128-131
The most effective dose of acetazolamide to prevent AMS has been debated in the literature. A systematic review done in 1999 concluded that at least 750 mg daily of acetazolamide was required and that the number needed to treat was 2.9 to prevent one case of AMS.123 The authors reported that 500 mg was no better than placebo. This review was criticized for not including all studies and for a bias towards studies using higher doses.133,134 Studies subsequent to this systematic review support a dose of 125 to 375 mg twice a day.135,136 In a well designed three arm trial, 162 trekkers ascending from 3440 to 4928 m were randomized to placebo, 125 mg, and 375 mg acetazolamide; no significant difference was found in AMS incidence between the two doses of acetazolamide; 24% and 21% respectively vs. 51% in the placebo arm.137 In conclusion, the best available evidence supports 125 mg PO bid as an effective dosing of acetazolamide for AMS prophylaxis.
The side effect profile of acetazolamide includes parasthesias, polyuria, myopia, nausea, and an adverse effect on the taste of carbonated beverages. Acetazolamide cannot be given to those who are sulfa allergic. Another side effect that needs to be considered is that acetazolamide has a negative effect on exercise, and at sea level it is known to reduce hypoxic work ability.138 This decrease in exercise ability, however, appears only to be an effect at sea level, not in a hypoxic environment. In a 1983 study, Schoene et al found that, in healthy volunteers at sea level, acetazolamide caused a decrease in VO2 max (the maximal oxygen uptake by an individual: a measurement of their maximal aerobic capacity) from 3.8 to 3.1 L/min while exercising in a state of normoxia, but caused an increase in VO2 max from2.4 L/min to 2.6 L/min in a hypoxic environment.139 These findings were confirmed in a 1986 placebo controlled study involving 21 subjects.140 A 2006 study showed that, while acetazolamide decreased muscle endurance at sea level, it did not do so at a chamber altitude of 4300 m (compared to placebo).141 Therefore, the literature supports the negative exercise effect of acetazolamide only at sea level but not at altitude.
Dexamethasone is the second most commonly used drug for altitude illness prevention. In a 1984 New England Journal article simulating altitude in a hyperbaric chamber, dexamethasone 4 mg PO qid starting 48 hours prior to exposure to 4570 m resulted in a significant decrease in AMS, from 60% in controls to 31% in the treatment group.142 When the drug was discontinued, those treated developed a progressive increase in AMS symptoms. Studies investigating lower doses of dexamethosone have not demonstrated benefit in AMS prevention.143,144 The number needed to treat in order to prevent one case of AMS with dexamethasone is reported to be 2.8.
In addition to its effect on the incidence of AMS, dexamethasone is also useful in reducing the negative cognitive effects of altitude exposure. At the standard dose of 4 mg every six hours, dexamethasone use was associated with improved cognitive performance, improved affect, and improved personality on acute altitude exposure. When stopped after three days, no adverse effects were seen in any of these categories.145
While dexamethasone is the best studied steroid for AMS prevention, other steroids have also been evaluated. In a small study of 40 patients, prednisolone 20 mg once a day two days before and three days at 3450 m reduced AMS, but did not effect the hypothalamic-pituitary-adrenal axis as measured by ACTH, cortisol, epinephrine, and norepinephrine levels.146 This finding is difficult to explain in that 3 mg of dexamethasone (the equivalent of 20 mg of prednisolone) was found to be ineffective, as mentioned above.
The combination of acetazolamide and dexamethasone has also been studied. Ellsworth et al carried out a double blind, placebo controlled, crossover study on 18 Mount Rainier climbers comparing acetazolamide, dexamethasone, and placebo. Acetazolamide and dexamethasone both significantly reduced AMS as compared to placebo, with dexamethasone being the most effective prophylactic agent for this type of rapid ascent (from sea level to 4500 m in an average of 33.5 hours).147 In a 1988 study, the combination of cetazolamide 250 mg PO bid and dexamethasone 4 mg PO qid was found to be superior to dexamethasone alone.149 A combination of acetazolamide (500 mg SR qd) plus low dose amethasone (4 mg bid) was found to be statistically more effective at preventing AMS over a two day ascent from 3698 to 5334 m as compared to the same dose of acetazolamide alone.149 Based on these studies, expert opinion now recommends this drug combination be utilized for unavoidable rapid ascents, such as rescue missions or military operations at high altitude.150
Antioxidants,151 clonidine,152 and theophylline153 have been studied to prevent AMS with varying levels of success . . . but none persuasive at this time.
Hyperbaric treatment has demonstrated temporizing benefit in the treatment of AMS. In a study of 64 patients, one hour of treatment at 193 mbar in a portable hyperbaric chamber led to significant short term improvement in AMS symptoms, but, by 12 hours, the benefit was no greater than symptomatic treatment alone.154 In a comparative study of hyperbaric oxygen to oxygen therapy, hyperbaric treatment was shown to be as effective as 4 L O2 when 24 patients were given two hours of either at 2850 m; the positive effect lasted at least one hour after treatment but no long duration follow-up was done.155
The best prevention of HAPE is slow ascent at a rate of no more than 300 to 350 m per day above 2500 m.156 Nifedipine 20 mg every eight hours or 30 to 60 mg a day of sustained release formulation can be utilized if a slow ascent is not possible in HAPE susceptible individuals.157 almeterol (125 mcg bid) has also been shown to be effective in reducing the incidence of HAPE in susceptible individuals.141 It has also been suggested that climbers drink red wine to prevent HAPE, though this has never been taken beyond a theoretic exploration!158
An October 2006 study found that dexamethasone significantly reduced the incidence of HAPE in susceptible individuals.159 In this study on HAPE prone subjects, 0/10 of those on 8 mg PO bid of dexamethasone developed HAPE vs. 7/9 on placebo. Dexamethasone may be effective for HAPE through a number of mechanisms: by decreasing pulmonary arterial pressure via enhancing the production of nitric oxide in HAPE prone individuals who otherwise have ecreased nitric oxide at altitude, 160 by increasing the clearance of fluid from the alveoli,161 and by increasing the productive of alveolar surfactant.162
There is limited literature on the treatment of HAPE. Textbooks provide recommendations, but supporting evidence is scarce.2-4,156 Oxygen and descent are the mainstay of any treatment strategy.
Mild HAPE (Table 5: Grade I and II) can be treated with bed rest with or without oxygen supplementation (2 to 4 L / minute by nasal cannula). This was first shown in a study by Marticorena and Hultgren in 1979 which compared treatment of mild HAPE with bed rest alone as compared to bed rest with oxygen. Bed rest alone resulted in resolution of mild HAPE in those so treated, but symptoms and vital signs (P < 0.01 for heart rate improvement) resolved quicker in the bed rest and oxygen therapy group. While this study showed that bed rest alone can be sufficient to treat mild HAPE, the authors recommended the use of oxygen whenever possible due to the more rapid improvement seen with its use. A retrospective review of patients treated in Summit County, Colorado (2800 m) showed this approach of oxygen and bed rest to be safe and efficacious; 25 of 58 patients seen with HAPE at two Summit County primary care centers were discharged to home or hotel on oxygen with ubsequent follow-up and good outcome. This study was limited by being retrospective, having no control group, and not providing any prospective criteria to determine who received outpatient treatment versus those who were admitted for care.163 Despite these limitations, this study did show that, for "mild" HAPE, bed rest and oxygen are effective treatments.
Moderate to severe HAPE may be treated with oxygen and one of several pharmacologic agents. Once the patient is in the ED and on oxygen, any other pharmacologic therapy is purely adjunctive and there are no controlled studies on adjunctive medication use. Pharmacologic adjuncts work through their effect on the elevated pulmonary pressures of HAPE. Table 11 provides a comparison of several pulmonary vasodilators and their efficacy in reducing pulmonary hypertension, with oxygen and phentolamine in combination giving the best results.164

Nifedipine: Nifedipine was first shown in 1989 to be an effective pharmacologic therapy for HAPE. 165 Nifedipine works both to treat HAPE without descent or oxygen and is being an effective prophylaxis in known HAPE susceptible subjects. For HAPE prevention, 20 mg tid is an effective dose if started on day of ascent and continued for 72 hours at altitude.166,167
-agonists have recently received interest in the treatment of HAPE due to their ability to enhance alveolar clearing of fluid. In a well designed study, salmeterol was shown to decrease the incidence of HAPE by 50% in HAPE susceptible individuals (from 74% in the placebo group to 33% in the salmeterol treated group).157 Albuterol has not been studied for the prevention or treatment of HAPE but is recommended as it should function similarly to salmeterol.168
Furosemide and morphine have been occasionally recommended for the treatment of HAPE.169,170 However, since the recognition that HAPE is not the result of vascular fluid overload, furosemide is no longer routinely recommended for HAPE; in addition, animal studies show an increase in mortality with this treatment.113 In one retrospective case review, patients who received furosemide had prolonged hospital stays compared to those who did not receive it. 26 It is unclear from this study if the negative effect of furosemide was a true cause and effect or rather an association with those who had more advanced HAPE being preferentially given diuretics. Several expert opinions have recommend against the use of furosemide (and morphine), at least in the prehospital environment and, if utilized at all, it is recommended that these medications be reserved for the hospital setting where blood pressure and volume status can be monitored.4,171,172
The future treatment of HAPE will likely involve sildenafil or other type-5 phosphodiesterase inhibitors. These inhibitors are the subjectof a significant amount of research due to their ability to reduce pulmonary artery pressure. In a 2005 study involving 12 subjects, sildenafil returned pulmonary artery pressures to normal and improved oxygen saturation as compared to placebo.173 In another 2005 study, sildenafil reduced pulmonary arterial pressure 12 mm compared to controls, and the six minute walk distance increased by 44 m compared to placebo controls in those taking 25 mg or 100 mg every eight hours; controls had no improvements. 174 In a study of 14 subjects tested at sea level while breathing 10% oxygen and then at an Everest base camp, sildenafil 50 mg PO increased exercise capacity.175 In a comparison of sildenafil to procardia, arginine, and acetazolamide, sildenafil was the most effective at lowering pulmonary arterial pressures in individuals without HAPE. In these individuals, nifedipine had minimal effect.176 A 2006 study by Olez et al found tadalafil, a longer acting type-5 phosphodiesterase inhibitor, to significantly reduce the incidence of HAPE in susceptible individuals
ascending rapidly to altitude.159
HAPE is treated with supplemental oxygen and descent. If adjunctive therapy is to be used, the literature support is good for the calcium channel blocker nifedipine, 10 mg PO followed by 20 mg of sustained release preparation every six hours.
Salmeterol (or albuterol) has been shown to be effective in preventing HAPE and, due to its ready availability and ease of use, has been ecommended as an adjunctive therapy in the ED. Other agents that appear effective as prophylaxis for HAPE, such as dexamethasone, tadalaphil, and sildenafil, have no studies or consensus ecommendations for use in the ED at this time.
There are no prospective, randomized studies on HACE treatment. As in all altitude illness, continued oxygen supplementation to maintain oxygen saturation greater than 90% is standard. All reviews ecommend dexamethasone 4 mg every six hours (8 mg loading dose if given in the field).2-4 Beyond these two therapies, other HACE care is supportive. In those cases where the patient is intubated, hyperventilation is not recommended in that they already have a respiratory alkalosis and further hyperventilation may worsen cerebral ischemia. Furosemide, mannitol, and oral glycerol have all been utilized, but there are no well designed studies to evaluate the effectiveness of these agents. Use of these agents is, at best, of indeterminate effectiveness. If these agents are utilized, care must be taken to ensure volume status is maintained.
This may be the most common condition seen at altitude.177 HAP/B was seen in 12% of Himalayan trekkers; this is 1.5 times the 8% incidence of AMS seen in the same group. In a series of climbers on Mount Aconcagua, 13 of 19 (68%) developed HAP/B.178 This condition has little literature written on it but is felt to be extremely common in those who spend over two weeks above 18,000 feet; it is felt to be a result of the hyperventilation of altitude and the very dry, cold air of altitude.179 This same review recommends that patients wear a face mask, suck on hard candy, and drink plenty of fluids to treat this condition. This condition can also cause severe coughing which may require symptomatic treatment. HAP/B can be confused with HAPE and vice versa; when treating HAP/B, one must ensure HAPE is not present.
This is a common condition at high elevations; 22 of 36 climbers who ascended to 5360 m developed HAR as manifested by retinal hemorrhages or "cotton wool" spots on exam.180 The incidence appears to also be related to height, as 19 of 21 climbers who ascended above 25,000 ft developed high altitude retinal hemorrhages.181 There is no specific treatment for HAR, other than descent if vision problems develop.
The effects of altitude on the eye are multifold; of greatest recent concern is the finding that those who had undergone Radial Keratotomy (RK), which involved making incisions in the cornea with a diamond blade, developed vision changes on exposure to altitude; fortunately those who had Photorefractive Keratectomy (PRK), which involves using a laser to create refractive changes in the cornea, did not develop vision changes.182 A 1998 study by Winkle found that all tested subjects who had undergone RK developed a significant hyperoptic shift and corneal flattening when exposed to ocular hypoxia. This study shows that patients who underwent RK are subject to vision changes at altitude and that hypoxia, rather than hypobaria, is the etiology. 183 The vision changes in those with RK takes 24 hours at altitude to occur and cannot be readily reversed while at altitude.184,185 The changes with LASIK, (which is now one of the most common refractive procedures utilized and involves utilizing a lazer to reshape the cornea under a cornel flap), even at extremely high altitude, are minimal. This was confirmed in a field study on Everest where six climbers who had undergone LASIK ascended Everest. Five were asymptomatic below 26,400 ft, one developed mild blurring at 16,000 ft. Two others developed mild blurring at 27,000 and 28,500 ft. All improved with descent.186
Altitude also adversely effects color vision. Healthy teenagers brought to moderate altitude (3000m) developed a significant number of color vision errors, most significant in the blue-yellow range.
Patients with stable coronary artery disease appear to do well at altitude. A review by Alleman et al in 1998 showed stable coronary patients with good exercise tolerance, no exertional ischemic symptoms, and an ejection fraction of greater than 50% to be at low risk at altitudes of 2500 to 3000 m. The greatest risk is during the first few days of altitude exposure when the patient is subject to the initial sympathetic surge that accompanies altitude exposure.188 It is recommended in this review that those with CAD going to much higher elevations acclimatize for several days first. A review of individuals with stable coronary artery disease and an ejection fraction of 39 6% were also found to have a good tolerance to altitude exposure.189
Those with borderline hypertension have been shown to have an increased incidence of AMS.190 While this small study by Ledderhos showed that those with hypertension have an increased incidence of AMS, altitude does not appear to increase the risk of medical consequences to those with controlled hypertension. Thirty-three subjects with hypertension attending a conference at 2500 m developed no symptoms related to their hypertension while at attitude. 191 In an epidemiological case series of 935 hypertensive patients, there was no increase in strokes or cardiac failure due to altitude exposure. 5
In a comparison study of six type I diabetics with 10 normal controls climbing to 8200 m, no difference was found in AMS scores, heart rate, blood pressure, or blood lactate. Diabetics did require increased use of insulin at altitude.192 One of the confusing problems to consider is that the symptoms of AMS (headache, lightheadedness, nausea) can make recog nition of hypoglycemia more difficult.193 The literature on diabetic ketoacidosis (DKA) at altitude is limited, but, in the Moore Study, two of 15 climbers developed DKA; both were taking acetazolamide. This was felt to be a possible co-factor in its onset. The risk benefit of acetazolamide use in diabetics must therefore be considered. The greatest concern for diabetics at altitude and medical providers caring for them is that all types of glucometers are inaccurate at altitude, both over and under estimating the glucose level; this has been proven in a multitude of studies. It is therefore recommended that two types of glucometers be carried at altitude as well as three times the number of test strips and supplies one would typically utilize. Insulin pumps have been shown to have no problems with function at altitude, provided they are protected from cold.194
Sleep disturbance is ubiquitous at altitude, with most new arrivals experiencing increased fragmented poor quality sleep, with frequent arousals due to periodic "cheyne-stokes" breathing, as well as a shift to lighter sleep levels.195 Several sleep agents have been studied for improving sleep at altitude. Zolpidem improved sleep at altitude, including time of onset of sleep, duration of sleep, and a decrease in arousal from sleep. There was no adverse affect on respiratory function.196 Zalephon was also effective, though not as effective as zolpidem.197 Low dose temazepam was also shown to stabilize breathing, improve sleep, and not lead to any oxygen desaturation during sleep. 198 Acetazolamide has been shown to improve oxygenation and decrease periodic breathing at altitude.199
Sextagenarians can safely climb 8000 m peaks provided they are in good health, moderate fitness, and experienced. This was shown by a case series of seven men and one woman from 54 to 63 years old that ascended to 8035 m on Gasherbrum II. Three of the eight had borderline hypertension and eccentric hypertrophy of the left cardiac ventricle.200
A study of tenth mountain division veterans with an average age of 68 found that the elderly acclimatized well to 2500 m elevation with a return to sea level performance after five days. Those with coronary artery disease that is well compensated at sealevel do well at altitude, although ischemia may be provoked at modestly lower myocardial work rates.201
Oxygen saturation measurement during light exercise at altitude has been touted as a method of estimating acclimatization. Individuals who have or will develop AMS have lower than expected oxygen saturation. 202 A large case series published as an abstract only gives expected baseline oxygenation at multiple points for ascents of Everest and Kilimanjaro.203 It has also been suggested that this holds for those who are not exercising. In a 1998 study of 102 subjects, an oxygen saturation lower than other individuals at the same elevation correlated strongly with the likelihood of developing AMS.204 A larger study in 2004 of 169 subjects did not find a significant correlation between AMS and decreased oxygen saturation.205 Thus, it remains indeterminate if those with oxygen saturations lower than expected for a given elevation are destined to develop AMS.
One of the areas of greatest controversy for pharmacologic prophylaxis of AMS involves the utility of Gingko. Gingko is a free radical scavenger, inhibits lipid peroxidation, preserves oxidative phosphorylation, and has numerous arterial and venous vasoregulatory activities in addition to being a strong antagonist of platelet-aggregation factor.206 Gingko may also work through several mechanisms involving nitric oxide metabolism.207-209 Animal studies have reported that Gingko at high doses protects rats from developing HAPE.210 The first human studies showed Gingko to be remarkably effective in preventing AMS. A French study involving 44 subjects found Gingko to be 100% effective in reducing AMS when given at a dose of 80 mg PO bid starting five days before ascent.211 A subsequent study of Gingko, 80 mg PO bid for 24 hours prior to altitude significantly reduced AMS compared to placebo and acetazolamide at an altitude of 3700 m.212 The next two studies on gingko found it did not significantly reduce AMS, though both showed a strong trend towards reducing AMS. In the first, Gingko 60 mg tid for one day prior to rapid ascent to 4205 m showed a strong trend towards reducing AMS, though it did not reach significance (7 of 12 of those using Gingko developed AMS vs. 13 of 14 using placebo, P=0.07).213 In the second study, Gingko 120 mg PO bid starting three days prior to exposure showed a strong trend to reducing incidence and severity of AMS, but it did not reach significance (P=0.07); in this study, 125 mg acetazolamide did significantly reduce the incidence of AMS.214 These latter two studies failed to answer the question; however, both were underpowered.
However, the largest and most recent studies have found Gingko to be no more effective than placebo for preventing AMS. In the largest study involving 614 trekkers, Gingko was given for three to four doses as 120 mg PO bid at an altitude of 4280 m and continued for 24 to 48 hours; it was found to be no better than placebo for reducing AMS.130 In the most recent study, Gingko was also no better than placebo.215 Unfortunately, it still remains unclear if Gingko has any positive effect on reducing AMS when taken prophylactically. One significant problem with the studies is that there is a potential lack of standardization of the compounds utilized. As Gingko is not a pharmaceutical, it remains unclear if the compounds utilized in the multiple studies are the same. This variability of active compounds in the studies may explain some of the outcome differences. Fortunately, Gingko has minimal side effects so, if utilized, there is little downside. While bleeding has been touted as a risk of Gingko utilization, this risk is limited to a small number of case eports.216-218 Larger, well designed studies have not found any increased risk of bleeding when Gingko is utilized in doses similar to those suggested for prevention of AMS. In conclusion, pending better research, Gingko does not have sufficient literature at this time to support its use in the prevention of AMS.219
Patients with AMS are generally treated symptomatically and managed as outpatients. Patients with HAPE, on the other hand, require careful evaluation and consideration for admission. If, after initial treatment, the patient has a normal room air saturation and does not have dyspnea at rest, they can be discharged to bed rest for 24 hours with close followup.3 If a patient can be made asymptomatic on low flow O2 (1 to 4 L / minute and an oxygen saturation of greater than 90%), they can be discharged to their quarters on O2, with close follow-up.3 Clear guidelines for admission are, again, limited in the literature. A composite of recommendations from various studies cited in this article is presented in Table 12.220 Once admitted, case series indicate that patients are discharged in 4 2 days.

Patients with HACE require admission and close monitoring for progressive cerebral edema and its complications. Recovery takes place from 2 to 21 days; 25% recover within 48 hours. Patients presenting with coma have a higher mortality and more prolonged hospital course.
1. "We can keep going."
Failure to stop ascending to altitude when symptoms develop will lead to the progression of illness and possible severe illness and morbidity. AMS will typically resolve over 24 to 48 hours if the individual does not ascend further once symptoms occur.
2. "We don't need to take drugs."
In cases where one must ascend quickly and the risk of AMS is high, prophylaxis should be utilized as this may prevent progression to the more severe illnesses of altitude.
3. "It's only mild HAPE."
One must consider the full differential in evaluating those in the ED who appear to have altitude illness. Altitude illness typically improves quickly with descent or oxygen; failure to improve should strongly suggest an alternate diagnosis.
4. "He's just tired."
The earliest sign of HAPE is fatigue and breathlessness out of proportion to the rest of a party at altitude or a sudden decrease in an individuals exercise ability. HAPE can be treated quickly if caught at this early stage.
5. "He's stumbling because he's tired."
An individual with ataxia at altitude should be considered to have HACE until proven otherwise.
6. "I never get altitude sickness."
While prior experience at altitude is a good guide to likelihood of subsequent illness, it is also true that anyone can become ill if they ascend high enough and fast enough.
7. "We have to go to sea level to get better."
Descent does not need to be significant to reverse altitude disease, 500 m for AMS and 1000 m for HAPE and HACE are all that may be needed for life saving descent.
8. "Dexamethasone will help me to acclimatize."
Dexamethasone is effective in reducing AMS in rapid ascents but has no effect on acclimatization, and symptoms may return if the drug is stopped at altitude. Acetazolamide does aid acclimatization.
9. "Failure to use oxygen."
Oxygen is the gold standard treatment for HAPE and HACE and must be utilized if available to keep oxygen saturations greater than 90%. Pharmaceuticals are adjuncts to oxygen use.
10. "Failure to ask about medication allergies."
Acetazolamide is a sulfa based drug and should not begiven to those with significant sulfa allergies.
With millions of sports enthusiasts going to high altitudes yearly, it is inevitable that the practicing emergency physician will eventually be required to consult or to treat the patient with altitude illness. The two case vignettes at the beginning of this article introduced the spectrum of altitude illness. In the first case, the "hangover" symptoms and time course were typical for AMS. The presentation was mild and management did not necessitate descent, though descent is always a consideration. Acetazolamide could be used to speed the acclimatization process. The second vignette represented a life threatening case of HAPE. The time course, occurring the second night at a new altitude, was classic. The symptoms of tachycardia, cyanosis, and bloody sputum classified the patient as having severe HAPE (Table 5: Grade IV) and was at a significant risk for mortality. Descent was mandatory and the evacuation needed to involve minimal exertion and cold exposure for the patient. He required supplemental oxygen as his primary therapy, which unfortunately was not available nor was hyperbaric therapy. Some benefit pending descent could have been provided with pulmonary vasodilation therapy, e.g., nifedipine, salmeterol or albuterol, and dexamethasone.

All altitude illness can be prevented by ascending slowly: less than 300 m a day after 2500 m. If this is not possible and altitude illness occurs, then the definitive treatment is descent and oxygen. Pharmacologic therapy plays its most prominent role in the prophylaxis against altitude illness when slow ascent is not possible. Acetazolamide and dexamethsone are the preferred drugs for AMS prophylaxis. Nifedipine is the present recommendation for HAPE susceptible individuals who will be ascending rapidly. Ongoing research and development is centered on improved pharmacologic therapy, including the use of hosphodieterase inhibitors, sildenafil, and the longer acting agents, such as tadalafil, to prevent HAPE in susceptible individuals as well as for treatment.
|
|
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
|
|
Evidence-based medicine requires a critical appraisal of the literature based upon study methodology and number of subjects. Not all references are equally robust. The findings of a large, prospective, randomized, and blinded trial should carry more weight than a case report.
To help the reader judge the strength of each reference, pertinent information about the study, such as the type of study and the number of patients in the study, will be included in bold type following the reference, where available. In addition, the most informative references cited in this paper, as determined by the authors, will be noted by an asterisk (*) next to the number of the reference.
Ian Wedmore; Brooks T. Laselle
March 1, 2007