
|
|
Skin and soft tissue infections (SSTIs) are a frequent emergency department (ED) presentation, representing 3% of all visits in 2005.1 Over the last decade, the number of ED visits for SSTIs has increased.1 The most common pathogen isolated from purulent infections is community-acquired methicillin-resistant Staphylococcus aureus (CA-MRSA)2; however, the outbreak of this strain is a relatively recent phenomenon. Of note, CA-MRSA has pathogenesis, strain characteristics, epidemiology, and clinical features that are quite different from those of hospital-acquired MRSA (HA-MRSA).
Infection and wound management in the ED requires that the clinician understand the implications of the emergence of CA-MRSA. Traditionally, skin infections have been treated with beta-lactam agents, either a cephalosporin (eg, cephalexin) or an antistaphylococcal penicillin (eg, dicloxacillin); however, these older treatment paradigms and regimens for common infections may no longer hold true. Additionally, current treatment recommendations of SSTIs are not clear-cut and are sometimes controversial. The conflicting evidence and expert opinions reflect the lack of current quality data to inform treatment decisions.3
The goal of this review is to provide an analysis of the current literature and recommendations regarding the diagnosis and management of CA-MRSA in the ED based on the best available evidence.
A 45-year-old man presents to the ED complaining of a painful lump on his right thigh that appeared yesterday and has become progressively larger and more painful. He denies any trauma and thinks he was bitten by a spider. He is feeling feverish but he reports no malaise or other systemic symptoms. About 6 months ago, he had a small abscess on his left leg that spontaneously drained and resolved; he has no other significant medical history. He denies any substance abuse, had a negative HIV test 3 months ago, and has no history of diabetes mellitus. On physical examination, the patient is afebrile, with normal vital signs. A fluctuant, raised lesion 5 cm in diameter with a surrounding 2-cm rim of erythema, induration, and tenderness is noted on the right anterior mid-thigh. The lesion does not extend to the groin. There is no crepitus or lymphangitic streaking of the leg. A simple incision and drainage of the abscess seems to be in order until the medical student asks about the quality of the evidence regarding timing of wound cultures, how and when antibiotics are indicated, when MRSA needs to be considered, and if 2 antibiotics are better than 1 (too many questions from one student, you think!).
CA-MRSA: Community-acquired MRSA
CAP: Community-acquired pneumonia
CDC: Centers for Disease Control and Prevention
C difficile: Clostridium difficile
CI: Confidence interval
ED: Emergency department
FDA: US Food and Drug Administration
HA-MRSA: Hospital-acquired MRSA
HIV: Human immunodeficiency virus
IDSA: Infectious Diseases Society of America
IV: Intravenous
MIC: Minimum inhibitory concentration
MRSA: Methicillin-resistant Staphylococcus aureus
MSSA: Methicillin-sensitive Staphylococcus aureus
PVL: Panton-Valentine leukocidin
S aureus: Staphylococcus aureus
S pyogenes: Streptococcus pyogenes
SSTI: Skin and soft tissue infection
This evidence-based review involved an extensive search of PubMed, MEDLINE?, and Web of Science ? databases, as well as the Cochrane Database of Systematic Reviews. A search of the PubMed database using the terms {community associated OR community acquired} AND {methicillin-resistant Staphylococcus aureus OR MRSA} resulted in almost 2000 abstracts. The following MeSH terms were also used:methicillin-resistant Staphylococcus aureus; skin diseases, infectious; staphylococcal skin infections; pneumonia, staphylococcal; emergency service, hospital; and emergencies. The Web of Science search identified articles containing citations of key publications, whereas a search for relevant guidelines was performed via the Agency for Healthcare Research and Quality's National Guideline ClearinghouseTM. An online search of the Infectious Diseases Society of America (IDSA) and Centers for Disease and Control and Prevention (CDC) websites was also performed, and a thorough review of the consensus guidelines and evaluation of their citations were undertaken.
The existing literature on CA-MRSA infections is limited in several ways. There are few quality data. Most studies are observational or small case series. There are few randomized clinical trials and much of the literature consists of expert opinion and review articles.
Nevertheless, 2 important United States consensus documents were available: the IDSA's Practice Guidelines for the Diagnosis and Management of Skin and Soft-Tissue Infections and the CDC's summary of an expert panel meeting: Strategies for Clinical Management of MRSA in the Community.4,5The 2005 IDSA guidelines were written just as CAMRSA was emerging as an important cause of SSTI; thus, they do not reflect the considerable changes that have occurred in the epidemiology of CAMRSA and management of SSTIs over the last few years. Similarly, the CDC's expert panel guidelines of 2006 are vague and limited because of the lack of controlled trials involving CA-MRSA.
Additionally, several expert review articles and their citations were evaluated.6-9 The most significant CA-MRSA study to date was a prospective observational study performed in 2004 by the EMERGEncy ID Net Study Group, which evaluated SSTIs in more than 400 patients from 11 geographically diverse EDs.2
Consensus guidelines for the treatment of MRSA in the United Kingdom―originally published in 2006, with an update published in 2009,―were also reviewed, including specific CA-MRSA recommendations by Nathwani et al in 2008.10 The UK guidelines have the same limitations as the American guidelines; additionally, there is difficulty extrapolating these recommendations to the United States, which has different epidemiology, antibiotic susceptibilities, and practice norms.10-12
The nomenclature and classification of SSTIs can be confusing, complicating the correct interpretation of existing studies and guidelines. Table 1 describes the clinical presentations of some frequently encountered SSTIs.

There is a lack of a standardized classification system for SSTIs in the literature which also makes the review of relevant studies difficult and potentially increasing confusion about the guidelines and recommendations for their treatment. Numerous classification systems have been proposed using a variety of variables: acute versus chronic; localized versus diffuse; primary (infection of healthy skin) versus secondary (infection complicating an existing skin disorder such as eczema); and necrotizing versus non-necrotizing. One system divides SSTIs according to the severity of the local infection, the presence or absence of systemic signs or symptoms, and the presence or absence of comorbidities.13
The US Food and Drug Administration (FDA) uses a SSTI classification system based on the presence or absence of complicating factors to categorize and register new antimicrobials.14,15 Uncomplicated SSTIs include impetigo, simple abscesses, furuncles, and cellulitis. The FDA defines as complicated those infections involving deeper soft tissue or requiring significant surgical intervention (eg, a major abscess, infected ulcer, or burn) and those occurring in the presence of significant underlying medical conditions that may complicate the response to treatment. SSTIs likely to involve anaerobic and gram-negative organisms (eg, perianal abscesses) are also considered complicated. See Table 2 for additional information.
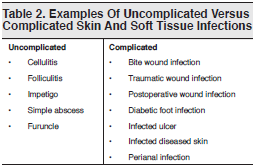
Adding to the confusion, the IDSA based its 2005 practice guidelines for SSTIs on specific conditions or disease entities (eg, cellulitis, necrotizing infection), mechanisms of injury (eg, bite, postoperative woundrelated infections), and host factors (eg, immunocompromised patients) rather than categorizing infections as uncomplicated or complicated.4 The IDSA guidelines were written just as CA-MRSA was emerging as a key pathogen; as a result, consideration of and coverage for CA-MRSA are not adequately emphasized in the document.
The outbreak of CA-MRSA is a relatively recent phenomenon. MRSA has long been recognized as a hospital pathogen, but it has only recently emerged as a community-acquired organism. MRSA isolates were first identified in 1961, shortly after penicillin was introduced into practice.16 Its prevalence increased slowly over the next several decades, primarily as a hospital-acquired (nosocomial) infection, HAMRSA. HA-MRSA typically causes serious invasive disease such as bacteremia, pneumonia, catheterrelated infections, and wound infections. The community-acquired strain, CA-MRSA, emerged in the mid-1990s. It causes primarily SSTIs but has also been implicated (although less commonly) in more invasive infections such as necrotizing pneumonia with sepsis.17,18
MRSA is now the most frequently isolated pathogen from SSTIs in patients presenting to the ED. In 1995, Frazee et al published a prospective case study using a convenience sample from a single ED in northern California. The data demonstrated a 51% incidence of MRSA in cultures obtained from these patients.19 The EMERGEncy ID Net Study Group's results, published in the New England Journal of Medicine in 2006, identified CA-MRSA as the most frequent pathogen isolated from SSTIs among ED patients.2 This study provided a snapshot of 422 patients with acute purulent SSTIs treated at 11 geographically diverse, metropolitan, universityaffiliated hospitals. S aureus was isolated from 320 patients (76%); of the S aureus isolates, 78% were CA-MRSA. The prevalence of CA-MRSA overall was 59% and ranged from 15% to 74% across the sites. MRSA was isolated from 61% of abscesses, 53% of purulent wounds, and 47% of patients with cellulitis with purulent exudate.
In a 2008 study published in Annals of Emergency Medicine, Pallin et al demonstrated that the number of US visits for SSTIs has increased markedly, rising from 1.2 million in 1993 (95% confidence interval [CI], 0.96-1.50 million) to 3.4 million in 2005 (95% CI, 2.8-4.1 million; P < .001 for trend).1 During the same period, the proportion of SSTI-related ED visits more than doubled, increasing from 1.35% in 1993 (95% CI, 1.07%- 1.64%) to 2.98% in 2005 (95% CI, 2.40%-3.56%; P < .001 for trend). Although the study used retrospective data from the National Hospital Ambulatory Medical Care Survey, these data represent a probability sample of visits to all US EDs (excluding Veterans Affairs, military, and federal hospitals). Visits identified as SSTI-related included diagnoses of cellulitis, abscess, furuncle, carbuncle, felon, impetigo, hidradenitis, folliculitis, and mastitis. This rise in ED visits for SSTIs coincided with the emergence of CA-MRSA. The number of ED visits for SSTIs almost tripled during this period, and physician behavior has changed as well. Notably, the number of prescriptions written by physicians for drugs active against CA-MRSA has increased.1
Historically, it was assumed that most SSTIs were caused by S aureus (ie, methicillin-sensitive S aureus, or MSSA) or Streptococcus pyogenes. Earlier conceptualizations of SSTIs further suggested that purulent infections (eg, abscesses) were more likely due to S aureus and nonpurulent versions (eg, cellulitis) were due to S pyogenes. This assumption may no longer be reliable, especially since the emergence of CAMRSA, particularly with regard to cellulitis (though data are limited).20 Since nonpurulent cellulitis cannot be adequately cultured, the incidence of MRSA as a cause is unknown. Limited data from older microbiologic analyses of cellulitis determined that streptococci (almost exclusively S pyogenes in erysipelas) were the most common pathogens associated with this infection; however, S aureus (ie, MSSA) has also been isolated.8,21
The molecular typing of MRSA reveals that CA-MRSA and HA-MRSA are different strains, with distinct genetic profiles, phenotypes, clinical characteristics, and antimicrobial sensitivity patterns.22,23
MRSA is defined as an organism with an oxacillin minimum inhibitory concentration (MIC) greater than or equal to 4 mcg/mL. Isolates resistant to oxacillin and methicillin are resistant to all beta-lactam agents (including penicillins and cephalosporins). Methicillin resistance is mediated by a penicillinbinding protein (PBP 2A) encoded by the mecA gene, which is located on a mobile genetic component called the staphylococcal cassette chromosome (SCCmec).20
Most isolates of CA-MRSA are genetically and phenotypically very similar. In CA-MRSA, the mecA gene encoding for beta-lactam resistance is almost always carried within SCCmec type IV, which is distinct from SCCmec types I, II, and III typically found in HA-MRSA. Pulsed-field gel electrophoresis has demonstrated that a few molecular types account for most cases of CA-MRSA.24 Most CA-MRSA is typified as USA300 (sometimes USA400), whereas HA-MRSA is USA100 or USA200. King et al demonstrated that all the MRSA isolates from outpatient SSTIs in Atlanta, Georgia, were a USA300 clone.25 In the EMERGEncy ID Net study involving geographically diverse hospitals, 97% of CA-MRSA isolates were the same USA300 clone.2
CA-MRSA appears to be more virulent than HAMRSA. Many CA-MRSA strains carry genes for Panton-Valentine leukocidin (PVL), a virulence factor associated with SSTIs and necrotizing pneumonia.26-28The PVL genes encode an exotoxin that confers virulence by creating pores in the leukocyte cell membrane and killing the cells. CA-MRSA is most often associated with the PVL-producing toxin, but exotoxins are not typical in HA-MRSA. In the EMERGEncy ID Net study, 98% of isolates were PVL positive.
HA-MRSA typically occurs in hospitals or other healthcare settings such as long-term care facilities and dialysis clinics. It causes serious invasive infections and is a typical pathogen in nosocomial pneumonias, especially in intensive care unit and ventilated patients. The pathogen is also a frequent culprit in postoperative wound infections and is frequently implicated in catheter-associated infections. The typical HA-MRSA patient is elderly, with multiple comorbidities, and has had a prolonged hospital course, whereas the typical CA-MRSA patient is young and previously healthy with no significant healthcare contacts. HA-MRSA is resistant to multiple antibiotics, unlike CA-MRSA, which is susceptible to many antibiotics, with the exception of the beta-lactams. Important differences between CA-MRSA and HAMRSA are summarized in Table 3.

The severity of the clinical presentation of the patient with CA-MRSA is dependent upon many factors, including the anatomic location of the disease and patient comorbidities, some of which interrelate with and influence each other.
The ED evaluation of the patient with an SSTI and potential CA-MRSA involvement must include a thorough history and targeted physical examination. The onset of the infection and any inciting injury should be elicited, as well as any symptoms suggesting systemic illness. Pertinent past medical history including comorbidities and immune status should be obtained, including any recent antibiotic use or hospitalization. Important physical findings include abnormal vital signs and a thorough evaluation of the location, type, and extent of the infection. (See Table 4)

For patients with an SSTI, the history should explore risk factors, potential etiology, and comorbidities.
Older expert opinion and guidelines tend to espouse consideration of CA-MRSA only in geographic locations where the prevalence rate is very high; however, recent data indicating a high prevalence of CA-MRSA over broad geographic regions makes this idea obsolete.2
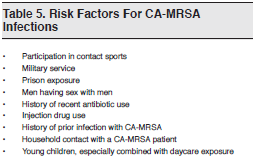
A number of risk factors for CA-MRSA infections have also been identified. (See Table 5) There have been many reports of CA-MRSA transmission in close-contact settings such as households,29,30 daycare centers,31,32 military facilities,33and prisons,34 as well as among athletes who participate in contact sport.35 Rates among men who have sex with men36 and among injection drug abusers37are also increasing.
Initially, it was thought that individuals who did not fall into a high-risk group were unlikely to have a CA-MRSA infection. Similarly, older expert opinion and guidelines suggested adding CA-MRSA coverage for infections only if patients met certain risk factors. However, 2 studies have demonstrated that risk factors and clinical judgment are unreliable at distinguishing between MSSA and CA-MRSA infections. In one study at a Los Angeles hospital, Miller et al collected clinical and microbiologic data on a consecutive series of patients with culture-proven S aureus infections and found that clinical and epidemiologic factors lacked the predictive capacity to distinguish patients with CA-MRSA infections from those with infections due to MSSA.38 A prospective observational study, meanwhile, found that an emergency clinician’s suspicion of CA-MRSA was a poor predictor of it.39 The study was conducted in an urban tertiary academic center and involved ED patients presenting with purulent wounds and abscesses from which cultures were drawn. Emergency physicians completed a questionnaire noting patient demographic data and their own suspicion for CA-MRSA infection. The prevalence rate of CAMRSA was 64%. Importantly, 76.4% of patients who were not considered at risk for CA-MRSA by the emergency clinician had culture-proven CA-MRSA. In this study, only intravenous (IV) drug use was significantly associated with the pathogen.
Many patients presenting with a CA-MRSA abscess initially report that they have been bitten by a spider.40,41 Patients with CA-MRSA-related infections often mistakenly believe their lesion is due to a spider or insect bite because of the spontaneous onset of redness and pain without an obvious injury.6,42 Clinicians may also mistakenly attribute the lesion to a bite, despite the fact the patient is in a geographic region where spiders capable of causing necrotizing wounds are not endemic.43,44 In one multicenter prevalence study conducted in 2006, patient reports of a spider bite as the etiology of a lesion correlated positively with CA-MRSA infection when compared with other bacterial etiologies.2
Comorbidities, particularly diabetes mellitus and immunosuppressive conditions such as human immunodeficiency virus (HIV), may adversely affect the response to an infection and increase the risk of complications. There are some studies in SSTIs that were conducted prior to the onset of CA-MRSA that evaluated risk factors and comorbid conditions that negatively impact outcome in SSTI.45,46
An assessment of the severity of an SSTI is considered essential in management decisions. The determination of severity should include evaluation of any systemic signs and symptoms and existing comorbidities and consideration of the anatomic location of the infection.6,47 For the most part, however, consensus guidelines and recommendations are vague and based on expert opinions with limited data to inform them.
The presence of delirium, lethargy, fever or hypothermia, tachycardia, and hypotension are crucial in assessing the seriousness of an infection.
Anatomic location is also important in the evaluation of an SSTI. Proximity to vital structures or joints makes an infection more serious (eg, an infection of the central face can lead to cavernous sinus thrombosis). Preexisting lymphedema or lymphatic damage may also impede resolution.
Numerous small, primarily observational studies have demonstrated good outcomes in patients with a purulent SSTI and documented CA-MRSA infection.48-52
In the large, geographically diverse EMERGEncy Net ID study, patients with purulent infections were followed up 15 to 21 days after the initial ED visit.2 In all, 96% reported that their infections had resolved. There were no significant differences in outcome between patients infected with CA-MRSA and those infected with other bacteria.
In the epidemiologic study by Pallin et al, the investigators found no increase in the proportion of patients admitted to the hospital, suggesting that SSTIs caused by CA-MRSA are no more serious than those previously encountered in the pre-CA-MRSA era.1 In an editorial that accompanied the study, Talan noted that while CAMRSA is relatively efficient at causing an SSTI, it is rarely associated with serious invasive disease and therefore should not be considered a “deadly superbug.” The author also suggested that CA-MRSA is often misinterpreted and sensationalized in the lay media.53
In an observational cohort study of 117 patients with SSTIs involving CA-MRSA and MSSA who were followed after discharge from hospitalization, no differences in long-term outcomes were identified, and both groups had similar nonresponse rates.54 Although outcome data are limited, the available evidence indicates that the majority of patients with a CA-MRSA-related SSTI have a favorable resolution.
Most clinical presentations of CA-MRSA involve relatively straightforward SSTIs, primarily abscesses and likely also purulent cellulitis; however, CA-MRSA has also been implicated as the etiologic pathogen in a number of other serious invasive clinical infections.55 For example, the strain has been reported as an etiologic agent in necrotizing fasciitis,56 septic thrombophlebitis,57pyomyositis, myositis,58,59 and sepsis.60-62 The incidence and role of CA-MRSA in diabetic foot infections is currently unknown.
Necrotizing fasciitis has traditionally been divided into monomicrobial and polymicrobial forms. S aureus is a known pathogen in the polymicrobial form, and now there are reports of CA-MRSA as a pathogen as well.56 Necrotizing infections are life and limb-threatening; unfortunately, their diagnosis is not straightforward. Clinical manifestations that suggest a necrotizing infection include abnormal vital signs, severe pain and/or pain out of proportion to the physical findings, rapidly advancing infection, crepitus, hemorrhage, sloughing, and blistering. These characteristics may not manifest until late in the course of the infection, however. (See Table 6.)

Pyomyositis is a deep abscess within striated muscle that is not due to local trauma or contiguous spread from adjacent tissue, but is usually associated with bacteremia in the setting of muscle injury. It is traditionally found in tropical climates, but cases due to CA-MRSA have been reported in the United States, particularly in immunocompromised patients.59
The current IDSA guidelines for SSTI treatment4 recommend that patients with signs and symptoms of systemic toxicity (abnormal vital signs such as fever or hypothermia, tachycardia, or hypotension) have a laboratory evaluation that includes:
The society also recommends that patients with abnormalities in these variables be admitted for aggressive evaluation and treatment, including surgical consultation, to exclude a deep invasive infection. (See Necrotizing Infections section, and Table 6.)

A risk stratification tool for predicting a necrotizing infection based on several laboratory parameters— the Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC)—has been developed. Points are assigned to different laboratory values and a total score is obtained. The maximum score is 13. A score greater than or equal to 6 has a positive predictive value of 92% for necrotizing fasciitis (95% CI, 84.3%-96.0%) and a negative predictive value of 96% (95% CI, 92.6%-97.9%). The Laboratory Risk Indicator has not been prospectively validated, and its ability to identify necrotizing fasciitis early in its course is unproven.63,64
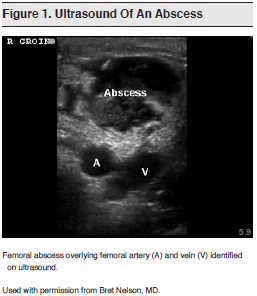
Ultrasonography can be a useful adjunct to help identify deep abscesses, differentiate cellulitis from an abscess, and guide drainage.65,66 (See Figure 1.)
No studies have answered this question, and expert opinion alone currently guides practice. Obtaining a culture during incision and drainage was considered unnecessary before the emergence of CA-MRSA.6Although many experts now advocate obtaining cultures routinely during incision and drainage,2,4,5,48 obtaining cultures for every purulent infection is controversial. Abrahamian et al have recommended not sending cultures when the results will not change management (eg, when antibiotics for CA-MRSA will be given regardless), pointing out that patients should not be billed for studies done solely for epidemiologic surveillance.7,67 Wallin et al have recommended sending cultures from the ED for a complicated abscess (defined by fever, lymphangitis, or significant surrounding cellulitis), for patients requiring admission, and for immunocompromised hosts.9
As mentioned previously, CA-MRSA’s role in nonpurulent SSTIs (ie, cellulitis without purulence) is unknown. Importantly, in the EMERGEncy Net ID study demonstrating a high prevalence of CA-MRSA, all of the patients had purulent infections for which cultures were easily obtained.2 Cellulitis generally cannot be cultured, and when a culture is drawn, it has low yield.8Yields for needle aspirations and more invasive punch biopsies in this disorder are also low and are generally not recommended.4
With cellulitis, blood cultures are positive in less than 5% of cases.68 Cultures are not routinely recommended for simple SSTIs but are recommended when there are more serious signs and symptoms or concerns about an invasive infection.4,8
Although HA-MRSA has been identified as a pathogen in nosocomial and ventilator-associated pneumonias for many years, it is not a community-acquired pneumonia (CAP) pathogen. CA-MRSA, on the other hand, can cause severe CAP. In 2002, Gillet et al published a report of 8 cases of CAP due to PVL-positive S aureus in France in which there were 6 fatalities.28 In recent years, reports of CA-MRSA causing serious CAP have increased.69-73 Many of these cases have involved previously healthy children and young adults.
Characteristic findings in CA-MRSA pneumonia include occurrence during an influenza outbreak or a preceding flulike illness and rapid progression to severe illness. The nationwide Emerging Infections Network identified 51 cases of CA-MRSA during the 2006-2007 influenza season.74 Of these, 47% of patients had a documented viral syndrome preceding identification, and 33% had confirmed influenza. The overall mortality rate was 51%.
Radiographic characteristics include patchy or homogeneous bronchopneumonia, cavitary infiltrates, pneumatoceles, pleural effusion, and empyema. Studies have identified the isolates as SCCmec type IV, USA300, and PVL positive for the exotoxin gene. Patients in these studies had high rates of respiratory failure and circulatory collapse as well as high mortality rates (25% to 60%).69-73 The possibility of CA-MRSA must be considered in any patient presenting to the ED with severe CAP. (See Table 7.)

Use of antibiotics, as well as choice and route of administration, should not only be determined by the likelihood of typical pathogens but should also take into account the type of SSTI and the severity of the clinical presentation (systemic signs, anatomic factors, patient comorbidities).
Antimicrobials should be prescribed judiciously, taking into account typical susceptibility patterns and incorporating regional or local antibiogram patterns. Antibiotics of choice have varied over time as resistance patterns have changed and additional options and data have become available. Unnecessary or indiscriminate use of antimicrobials with a broader range than necessary contributes to the development of antibiotic resistance and increases an individual’s risk of developing an infection with a resistant organism.
Consensus guidelines for treatment are vague, reflecting the lack of quality prospective data. In addition, available studies use inconsistent inclusion criteria and terminology, making interpretation of their results problematic.
Oral agents with activity against CA-MRSA include trimethoprim-sulfamethoxazole (TMP-SMX), clindamycin, tetracyclines, and linezolid. Currently available parenteral agents include vancomycin, linezolid, daptomycin, tigecycline, and telavancin. (See Table 8.)

The antibiotic TMP-SMX is commonly used to treat CA-MRSA infections in outpatients and is an especially important antibiotic option in uncomplicated SSTIs. It is not FDA-approved for the treatment of any staphylococcal infections. Virtually all CAMRSA isolates are sensitive to TMP-SMX; however, outcome and efficacy data are limited. In vitro susceptibility testing revealed that 95% to 100% of CAMRSA strains are susceptible to TMP-SMX.2,27,48 One in vitro study of the susceptibility of CA-MRSA to commonly prescribed oral agents demonstrated that TMP-SMX is rapidly bacteriocidal and was significantly more effective against MRSA compared with other orally available agents, including clindamycin, minocycline, and linezolid.75 Clinical efficacy data, however, are limited to observational and retrospective studies.50,51,76-78
No data indicate that TMP-SMX is active against S pyogenes. This is an important consideration in patients with cellulitis and complicated SSTIs. Data to support IV TMP-SMX use in more severe infections are also lacking.6
The optimal dosage of TMP-SMX is uncertain. No prospective studies are available to guide recommendations. One double-strength tablet contains 160 mg of trimethoprim and 800 mg of sulfamethoxazole. Some experts recommend 1 double-strength tablet twice daily, and others recommend 2 doublestrength tablets twice daily to optimize tissue concentration; however, higher doses may increase the incidence of adverse effects.79,80 TMP-SMX is a pregnancy category C drug and is not recommended for women in the third trimester of pregnancy or for children younger than 3 months. Adverse reactions associated with TMP-SMX include hematologic suppression (rarely) and Stevens-Johnson syndrome.
Clindamycin is an important antibiotic option for the treatment of SSTIs and is commonly prescribed for CA-MRSA. It is FDA-approved for the treatment of serious infections due to S aureus, though not specifically approved for MRSA. It has been used successfully to treat invasive CA-MRSA infections in children and is capable of inhibiting bacterial toxin production, including PVL.81
The susceptibility rate of CA-MRSA to clindamycin is not as high as its susceptibility rate to TMPSMX. A 2007 study in one Boston outpatient facility revealed that nearly half (48.2%) of CA-MRSA isolates were clindamycin-resistant.50 Emergency clinicians should be aware of their regional clindamycin susceptibility profile, since CA-MRSA susceptibility variation by geographic region has been reported.
Importantly, antibiotic susceptibility tests indicating that an isolate is susceptible to clindamycin may be misleading, as some strains of CA-MRSA (phenotype iMLSB) have demonstrated an inducible resistance to the drug. Resistance to erythromycin is a marker for inducible resistance to clindamycin.82A specialized laboratory test, the erythromycinclindamycin D-zone test (named for its characteristic shape on agar media) is recommended for detection of inducible clindamycin resistance in erythromycin- resistant/clindamycin-susceptible isolates.83-85Nevertheless, data regarding the clinical significance of inducible clindamycin resistance are conflicting.86
Clindamycin does have an advantage over TMPSMX in that it provides better coverage of S pyogenes, making it a monotherapy option for treatment of cellulitis (see Cellulitis). Clindamycin is an important drug in the armamentarium against CA-MRSA and offers advantages over TMP-SMX in some clinical scenarios. However, due to concerns over possible resistance, some experts discourage use of clindamycin as monotherapy in moderately or severely ill patients.6
The recommended adult dosage of clindamycin is 150 to 450 mg orally every 6 to 8 hours. The IV dose is 600 to 900 mg every 8 hours.79,80 Clindamycin is classified as pregnancy category B.
Diarrhea is a common adverse effect and is infrequently associated with Clostridium difficile diarrhea. Observational data suggest that C difficile may occur more often with use of clindamycin than with use of other antimicrobials.87
Long-acting tetracyclines used to treat SSTIs include doxycycline and minocycline. Doxycycline is FDAapproved for S aureus infections, but it is not specifically approved for MRSA.
Data supporting the use of these agents for CAMRSA are limited to observational and retrospective studies. In one small case series, doxycycline and minocycline appeared to be effective treatments for SSTIs caused by tetracycline-susceptible strains of MRSA.88 Effectiveness was also seen in a retrospective cohort study of outpatients with uncomplicated skin infections.89
Susceptibility of CA-MRSA to tetracyclines varies geographically, and a long-acting tetracycline is a reasonable option for an infection if local sensitivity profiles indicate high susceptibility. Doxycycline and minocycline are effective in the treatment of superficial SSTIs, but data are lacking to support use in more invasive infections. As with TMP-SMX, efficacy against S pyogenes is questionable, so their use in nonpurulent cellulitis is not recommended.6,8
The recommended dosage of doxycycline or minocycline is 100 mg twice daily.79,80 Tetracyclines are pregnancy category D drugs and are contraindicated in pregnant women and children because of deposition in teeth and bones.
Rifampin should never be used alone for the treatment of CA-MRSA. Although CA-MRSA often demonstrates susceptibility to the drug in in vitro profiles, resistance develops rapidly during treatment.90Rifampin is sometimes used as adjuvant therapy in complicated invasive MRSA infections, although data supporting this use are limited.77 Additionally, there is in vitro evidence of an antagonistic effect when rifampin is combined with TMP-SMX.75
Fluoroquinolones should not be used to treat SSTIs due to MRSA. Many MRSA strains have been shown to be fluoroquinolone-resistant, and even in susceptible strains, resistance can develop rapidly.2,48,91None of the fluoroquinolones are FDA-approved for the treatment of MRSA infections; however, ciprofloxacin and levofloxacin are FDA-approved for the treatment of complicated skin infections while ciprofloxacin, levofloxacin, and moxifloxacin are FDA-approved for the treatment of uncomplicated skin infections due to S aureus in adults.
Erythromycin, clarithromycin, and azithromycin are all FDA-approved for the treatment of uncomplicated skin infections. Due to high rates of resistance, however, these drugs should not be used for CAMRSA.2,48
Vancomycin is the standard of care for patients with MRSA requiring parenteral treatment and hospitalization; however, S aureus strains with reduced susceptibility to vancomycin have emerged, and there have been increasing MICs for S aureus (the socalled “MIC creep”).92,93 In addition, vancomycin is inferior to beta-lactams for S aureus bacteremia and endocarditis, and some data suggest inferior efficacy compared with antistaphylococcal beta-lactams for MSSA. Other data suggest that linezolid may be more efficacious for CA-MRSA-related pneumonia. Tissue penetration is variable.
The recommended vancomycin dosage is 15 to 20 mg/kg (every 8-12 hours if renal function is normal; adjusted dosing interval if the patient has reduced creatinine clearance rate); the maximum dose is 2 g. In critically ill patients, a loading dose of 25 to 30 mg/kg can be used. After patients are admitted, measurements of vancomycin trough levels subsequently inform the dosage.79,80,94 Vancomycin is classified as pregnancy category C.
In general, the alternatives to vancomycin (linezolid, daptomycin, tigecycline, and telavancin) are not recommended for empiric use in the ED. They should be reserved for use in patients who are unable to tolerate vancomycin or in patients with infections with known or suspected resistance to vancomycin.
Linezolid is FDA-approved for the treatment of SSTIs and nosocomial pneumonia due to MRSA in adults and children. S aureus resistance is rare, and when it does occur, it is associated with long-term use of the drug.
In addition to being active against virtually all CA-MRSA isolates, linezolid is active against S pyogenes. Linezolid also has the ability to suppress toxin production.
Linezolid appears to penetrate pulmonary tissues and lung epithelial lining fluid more effectively than vancomycin and may therefore offer an advantage over the latter. There are no head-to-head studies of vancomycin versus linezolid in treating CA-MRSArelated CAP, but there are studies comparing the 2 drugs in the treatment of HA-MRSA in ventilator and nosocomial pneumonias. Although efficacy data for use of linezolid in pneumonia are positive, there is concern about its use in endocarditis because it lacks bacteriocidal activity (this activity is important in treating endovascular infections).
The recommended dosage for linezolid is 600 mg twice daily for both oral and IV formulations. Linezolid has excellent oral bioavailability79,80 and is a pregnancy category C drug. Adverse effects include myelosuppression and peripheral and optic neuropathies (mostly with prolonged use). Linezolid is a weak inhibitor of monamine oxidase and has been associated with serotonin syndrome in patients on selective serotonin reuptake inhibitors. Linezolid is also very expensive compared with other orally available drugs for CA-MRSA.
Daptomycin is FDA-approved for complicated SSTIs in adults and children and for S aureus bacteremia and right-sided endocarditis.95 Daptomycin is inactivated by pulmonary surfactant and thus should not be used for pneumonia.96 Resistant strains can emerge during therapy. There may be cross-resistance with vancomycin; exposure to vancomycin may affect bacterial susceptibility to daptomycin due to cell wall changes.
The recommended dosage for daptomycin is 4 mg/ kg IV daily. Higher doses of 6 mg/kg are recommended for treating bacteremia and endocarditis.79,80 Daptomycin is classified as pregnancy category B. An important side effect is muscle toxicity.
Tigecycline is a tetracycline derivative that is FDAapproved for SSTIs due to MRSA and MSSA and for intra-abdominal infections caused by MSSA in adults. The recommended dosage of tigecycline is 100 mg IV 1 time, then 50 mg IV every 12 hours.80,97The drug is pregnancy category D and is not recommended in children younger than 8 years. Tigecycline is contraindicated in pregnancy and children due to potential deposition in teeth and bones.
Telavancin was recently approved by the FDA for treatment of complicated SSTIs in adults.98 It is a pregnancy category C drug and has been associated with serious nephrotoxicity.
Incision and drainage is the primary treatment for skin abscesses and furuncles. Adequate incision and drainage with thorough exploration for loculations is important for resolution of the abscess. Packing of the wound is usually recommended; however, a recent small clinical pilot study involving 48 patients has challenged the dogma that packing is necessary. Results showed similar outcomes in patients who did and did not receive packing, with less pain experienced by the latter group.99
Prior to the emergence of CA-MRSA, the recommended treatment for uncomplicated abscesses was incision and drainage alone, without the need for antimicrobials. Several older studies corroborated the efficacy of this approach, demonstrating no benefits with the addition of antibiotics.100-103 Despite this evidence, many practitioners have continued to prescribe beta-lactam antibiotics for abscesses after incision and drainage.104
Data on the use of antibiotics to treat abscesses due to CA-MRSA are limited. No study has specifically examined the impact of comorbidities or cellulitis. Nevertheless, available data—mostly from observational or small studies—suggest that antibiotics are unnecessary for the treatment of uncomplicated or simple skin abscesses, even in the era of CA-MRSA.
Expert opinions and treatment recommendations, including The Sanford Guide to Antimicrobial Therapy, state that incision and drainage alone is likely sufficient for abscesses smaller than 5 cm. This size cutoff is based on only one small study: a prospective observational study by Lee et al involving 69 children with abscesses that grew CA-MRSA. Results indicated that incision and drainage alone failed to resolve infected sites greater than 5 cm in diameter.49 The incision and drainage procedure was not standardized across the study, so it is possible that inadequate procedures and not abscess size or use of antibiotics was the culprit in treatment failure. In addition, the use of “less than 5 cm” as the definition of a “small” abscess not requiring antibiotics may be inappropriate for some bodily locations.
Several small retrospective studies of simple abscesses caused by CA-MRSA found no differences if a beta-lactam drug or drug active against CA-MRSA was used for treatment, suggesting that incision and drainage alone is sufficient.37,46,105 A retrospective cohort study of 441 abscesses due to CA-MRSA and MSSA found full resolution in 99.6% of patients treated with discordant antibiotics and in 98.8% of those treated with concordant antibiotics.106 In the prospective observational study by Lee et al discussed previously, 94% of subjects treated with discordant antibiotics improved.49
Treatment information available for 406 patients in the EMERGEncy ID Net study by Moran et al also suggests that incision and drainage alone is sufficient to cure MRSA infections. In the study, 19% of patients had incision and drainage alone, and 66% received both incision and drainage and antibiotics (most commonly the beta-lactam agents cephalexin and dicloxacillin). In 100 of 175 MRSA infections for which antibiotic treatment was provided (57%), antibiotic therapy was discordant with the results of susceptibility testing; however, there was no difference in outcomes between patients whose MRSA isolate was discordant and those in whom the isolate was concordant with the prescribed antibiotic.2
In a prospective, randomized, double-blind singlesite study by Rajendran et al of uncomplicated skin abscesses, 87.8% of which were identified as CA-MRSA isolates, cure rates after incision and drainage were 84.1% in patients who also received cephalexin and 90.5% in those who received placebo. The high cure rates again suggest that incision and drainage alone is sufficient.52Unfortunately, there was no treatment arm receiving a drug active against CA-MRSA, so any potential improvement in outcome could not be determined.
A small difference in outcome with the addition of an antibiotic effective against CA-MRSA was found in a retrospective cohort study of 531 patients by Ruhe et al.50 Most patients underwent incision and drainage. Treatment failure occurred in 45 of these patients (8%). A significant difference was found in rates of treatment failure, with a 5% failure rate for those receiving antibiotics effective against CA-MRSA and a 13% failure rate for those receiving antibiotics ineffective against CA-MRSA. Use of a CA-MRSA-inactive antibiotic was an independent predictor of treatment failure (adjusted odds ratio, 2.80; 95% CI, 1.26-6.22). A second retrospective review of 399 patients at a single outpatient site in Boston also found that the use of a concordant antibiotic was associated with increased likelihood of clinical resolution (adjusted odds ratio, 5.91) when controlling for incision and drainage and HIV status.51A review of available evidence by Hankin in 2007 supports that incision and drainage alone for simple abscess is adequate.107
Determining which abscesses will benefit from empiric antibiotic coverage is an area of controversy that has not been specifically studied.108 Guidelines from CDC and IDSA regarding this question are based primarily on expert opinion.4,5 Factors integral to deciding whether to add empiric antimicrobial coverage after an incision and drainage procedure include associated cellulitis; lymphangitis; signs and symptoms of systemic illness (eg, fever, hemodynamic instability); associated comorbidities such as diabetes mellitus, HIV, and other immunosuppressive states; extremes of age; location of the abscess (eg, proximity to joints or vital structures, areas that may be difficult to drain completely, and areas prone to complication such as the central face); and lack of response to initial incision and drainage (although the drainage may have been inadequate or further incision and drainage may be needed).
One retrospective single-site study of 50 consecutive cases with a high prevalence of CA-MRSA measured compliance with a treatment algorithm based on categorization of SSTIs into 3 arms: cellulitis/ folliculitis, simple abscess, and complicated abscess. The failure rate was only 3% when treatment conformed to the algorithm, but it climbed to 62% when the algorithm was not followed. The study design selected for complicated infections with significant comorbidities.109
Data are needed to determine whether adding antibiotics limits the progression to more severe or invasive disease, decreases the time to resolution of the infection, and decreases the rate of recurrence (and the subsets of patients in which this occurs). See Table 9, for factors that should be taken into account when considering whether to add antibiotics to the management of abscesses.

Prior to the emergence of CA-MRSA, the usual practice was to treat SSTIs with drugs that have good activity against MSSA and S pyogenes, typically penicillinase- resistant semisynthetic penicillins such as dicloxacillin or with first-generation cephalosporins such as cephalexin. This practice is no longer acceptable in the era of CA-MRSA. Purulent infections have a high likelihood of CA-MRSA, and treatment should include an antimicrobial agent that is active against it. When there is an abscess with cellulitis, the decision to also cover for S pyogenes is murky. With purulent cellulitis, CA-MRSA should probably be covered. Note that the literature contains no detailed descriptions of what constitutes an abscess with cellulitis versus cellulitis with purulent drainage.
Cellulitis is likely to be due to S pyogenes, and treatment should include an antimicrobial agent that is active against this organism. As mentioned previously, CA-MRSA’s role in nonpurulent SSTIs is unknown. The decision to cover for both S pyogenesand CA-MRSA in uncomplicated cellulitis without purulent drainage is controversial, with little guiding data.
Coverage for both CA-MRSA and S pyogenes should be considered in the following scenarios:
Possible choices for covering both CA-MRSA and S pyogenes include the following agents:
Ideally, patients should have a follow-up reevaluation of their infection within 24 to 48 hours of treatment to verify the clinical response.4,5 There are no studies to guide admission decisions. Expert opinion, as well as common sense, suggest that patients should be admitted if they have a SSTI accompanied by signs or symptoms of systemic toxicity.
The IDSA guidelines recommend that treatment for uncomplicated cellulitis should continue for 5 days. One study has demonstrated this duration is as effective as a 10-day treatment course.110 Other expert opinions suggest that the clinical response should guide the duration of therapy. This may not be practical for many ED patients with limited access to outpatient follow-up care, however.
Coverage for complicated SSTIs should include an agent that is active against CA-MRSA. Parenteral treatment should be considered for extensive soft tissue infections with signs of systemic illness including fever or in the presence of diabetes mellitus or an immune deficiency disease.
Vancomycin is the standard of care for moderate to severe infections. Clindamycin may also be considered, because it covers S pyogenes and some anaerobes, and also offers the advantage of decreasing exotoxin production. Emergency clinicians should be aware of local resistance patterns, however, as well as the possibility of inducible resistance. Due to potential resistance problems, many experts do not recommend clindamycin as monotherapy for complicated SSTIs or invasive infections.
Linezolid has been shown to be as effective as vancomycin for SSTI.111 One study demonstrated that linezolid is preferable for treating MRSA in complicated SSTIs,112 although another study disputed this finding.113
Linezolid, daptomycin, and tigecycline have demonstrated efficacy in prospective controlled trials involving serious MRSA infections.114 The agents have not been studied in head-to-head trials, however, so their relative efficacies are unknown.
The incidence of CA-MRSA in diabetic foot infections is unknown, but current expert recommendations (eg, The Sanford Guide to Antimicrobial Therapy) advocate including CA-MRSA coverage.80The current IDSA guidelines for diabetic foot infections were written before the dramatic rise in CAMRSA infections and recommends coverage in areas with a high prevalence of the bacterial strain.115
Necrotizing infections require prompt recognition, critical care resuscitation, surgical debridement, and broad-spectrum antibiotics. Empiric antimicrobials should include vancomycin for coverage of MRSA. The inclusion of an agent capable of inhibiting toxin formation (eg, clindamycin or linezolid) is also believed to be of benefit. The Sanford Guide to Antimicrobial Therapy discourages adding linezolid to vancomycin because of a potential antagonistic interaction.80
The current IDSA/American Thoracic Society guidelines for treatment of severe CAP include the addition of vancomycin or linezolid for coverage of suspected CA-MRSA.116 High failure rates for MRSA in nosocomial infections treated with vancomycin have been attributed to its poor penetration into lung tissue. Linezolid is a viable alternative to vancomycin, but no head-to-head studies have compared the drugs’ efficacy in treating CAP. Several studies of nosocomial HA-MRSA have informed recommendations for CA-MRSA-related pneumonia in the absence of specific clinical trials. A retrospective, pooled subgroup analysis found higher cure rates and improved survival with linezolid versus vancomycin,117 although 2 prospective studies showed comparable cure rates for the drugs,118,119 and a randomized study comparing them found no significant differences.120 Daptomycin is inactivated by pulmonary surfactant and should not be used for pneumonia.
Wallin et al has recommended adding clindamycin or linezolid to treatment for life-threatening infections due to their ability to counteract toxin production. The CDC’s CA-MRSA guidelines suggest consulting with the hospital infectious diseases department regarding linezolid use in this situation.5
Contagion of CA-MRSA among close contacts is well-known. (See Risk Factors section and Table 6) A prospective cohort study that followed patients after hospitalization with a CAMRSA infection demonstrated a trend for close contacts of patients to develop similar skin infections.54The CDC’s CA-MRSA guidelines recommend several basic hygiene measures aimed at preventing such spread.5 (See Table 10.)


The role of fomites in the transmission of CAMRSA is unclear. It is well-known that HA-MRSA can survive for prolonged periods, possibly weeks, on inanimate objects. It is unknown if the same holds true for CA-MRSA.
Decolonization of S aureus refers to the use of antiseptics or antimicrobials to eliminate or suppress carriage of the pathogen and prevent re-infection or transmission. Most available data regarding S aureus decolonization are in HA-MRSA, not CA-MRSA. According to the CDC, no data support the use of agents such as nasal mupirocin and antiseptic body washes (eg, chlorhexidine) to eliminate S aureus colonization.5 Although the optimal management of recurrent CA-MRSA infections and the benefits of decolonization are unknown, 2 potential (but inadequately studied) decolonization strategies have been suggested: the use of either topical chlorhexidine gluconate or diluted bleach. Whether or not topical chlorhexidine gluconate should be left on the skin before rinsing―and for how long―is not known. For decolonization with bleach, the recommendation is to use 1 teaspoon (3.4 g) of bleach per 1 gallon (3.8 L) of bath water.121 Oral antimicrobials are not recommended for decolonization of nosocomial S aureus infections.122,123
CA-MRSA is the most frequently isolated pathogen from purulent SSTIs in ED patients. An almost 3-fold increase in ED visits for SSTIs coincided with the rise in CA-MRSA infections.1 CA-MRSA differs from HA-MRSA and has distinct strain characteristics, pathogenesis, clinical manifestations, and antibiotic susceptibility patterns. The emergency clinician's suspicion of MRSA infection is a poor predictor of its incidence because clinical features and risk factors cannot reliably distinguish SSTIs caused by CA-MRSA from those caused by MSSA.
It is crucial that the emergency clinician understand the impact of CA-MRSA on the management of SSTIs. More data from controlled clinical trials are needed to establish optimal regimens for the treatment of SSTIs in the age of CA-MRSA. Most simple abscesses in healthy hosts are cured by incision and drainage alone; however, when abscesses are complicated and require antibiotic therapy, an agent that covers CA-MRSA should be included. The role of CA-MRSA in nonpurulent cellulitis is unclear.
Optimal antibiotic regimens for the treatment of SSTIs are undetermined, but current oral antibiotic options with CA-MRSA activity include TMP-SMX, clindamycin, doxycycline or minocycline, and linezolid. Of these, only clindamycin and linezolid have good S pyogenes coverage. When using agents that are not active against this pathogen, additional coverage for cellulitis or complicated infections should be considered. Emergency clinicians must be aware of local susceptibility patterns and, in the case of clindamycin, the possibility of inducible resistance. For ill patients with a complicated SSTI or systemic illness requiring hospitalization, vancomycin remains the drug of choice. Several other parenteral options are available, including linezolid. Although CA-MRSA most often causes easily treatable SSTIs, it is notably―(but rarely)―implicated in serious invasive illnesses, including necrotizing infections. Empiric treatments for severe CAP must include MRSA coverage, either with vancomycin or linezolid.
An incision and drainage of the abscess was performed and purulent material was drained. The wound was explored for loculations, irrigated with saline, and packing was placed. A specimen for culture and sensitivity testing was sent to the microbiology laboratory. Due to the large size and surrounding area of cellulitis, the patient was placed on antibiotic coverage with TMPSMX, 1 double-strength tablet twice daily. Hygiene measures that might decrease the risk of transmission to his household members were discussed, including keeping the wound covered and thorough hand washing. He returned as instructed in 2 days for a wound check and packing removal; the infection was improving. Culture of the wound was positive for MRSA sensitive to TMPSMX. The patient asked if anything could be done to prevent future similar infections. He was informed that decolonization treatments with oral or topical agents do not have any proven benefit. However, hygiene practices such as not sharing razors or any equipment that comes in contact with skin as well as the potential benefit of using a bleach-containing solution for cleaning household surfaces were recommended.











Evidence-based medicine requires a critical appraisal of the literature based upon study methodology and number of subjects. Not all references are equally robust. The findings of a large, prospective, randomized and blinded trial should carry more weight than a case report.
To help the reader judge the strength of each reference, pertinent information about the study, such as the type of study and the number of patients involved, are included in bold type following each reference. In addition, the most informative references cited in this paper, as determined by the authors, are noted by an asterisk (*) next to the number of the reference
Denise Nassisi
October 1, 2010