Table of Contents
About This Issue
Hypernatremia and hyponatremia can present similarly, with often-vague complaints such as weakness, nausea, vertigo/falls, abdominal pain, tachypnea, confusion, and seizure. Patients who are unable to regulate their water intake (eg, infants, the elderly, and those with mental or physical impairment) and those with chronic diseases (heart, kidney, liver, cancer, and drug/alcohol use) are most at risk. In this issue you will learn:
The risk factors, history, and symptoms for hypernatremia and hyponatremia.
The levels of serum sodium concentration that will point to the etiology of the disturbance.
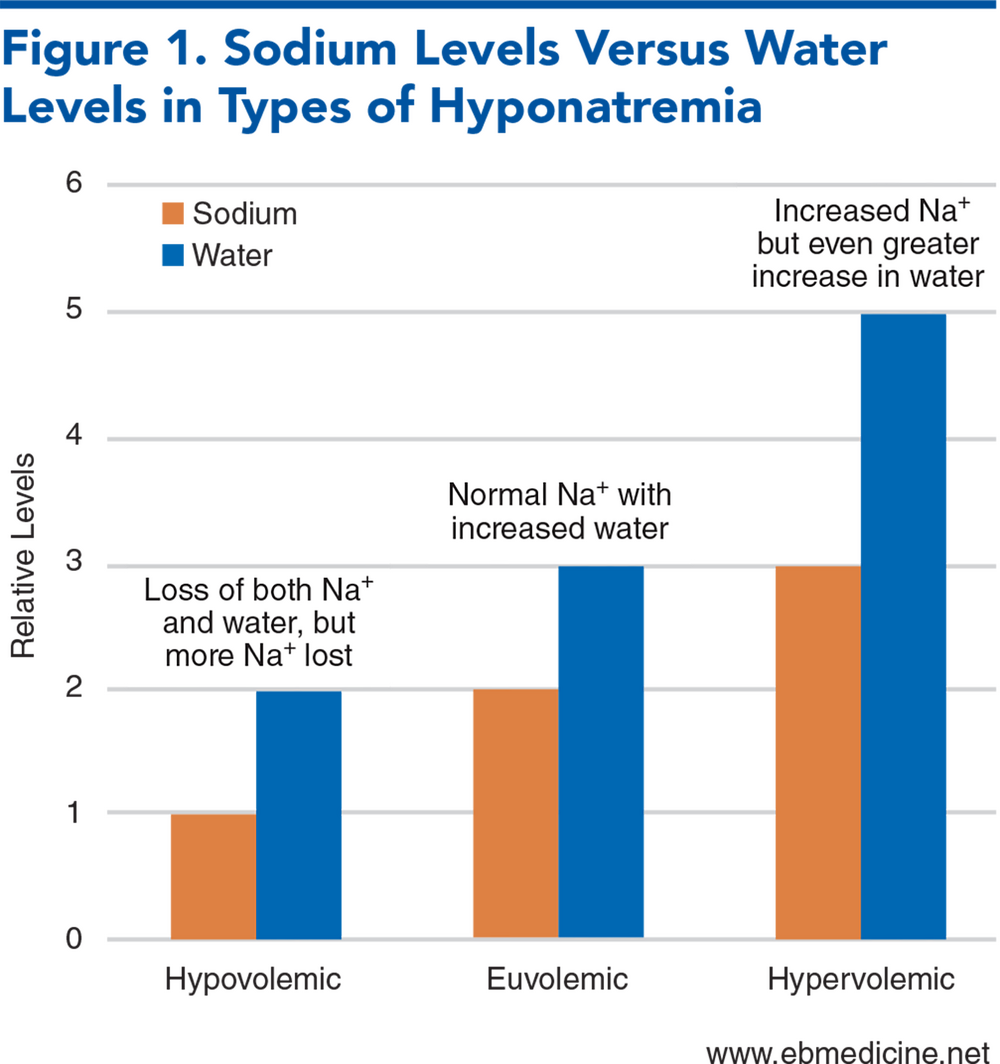
The different likely causes of hypovolemic hyponatremia, euvolemic hyponatremia, and hypervolemic hyponatremia.
How to distinguish between pseudohyponatremia and hyponatremia.
The role of diabetes insipidus and syndrome of antidiuretic hormone secretion (SIADH) in determining sodium imbalances.
Important equations for calculating total body water deficit and the effect of infusates on serum sodium.
Which IV fluids to use, which to avoid, and how quickly electrolyte disturbances should be corrected.
How to differentiate exercise-associated hyponatremia from dehydration.
- About This Issue
- Abstract
- Case Presentations
- Introduction
- Critical Appraisal of The Literature
- Etiology and Pathophysiology
- Volume and Osmolarity Regulation: The Simplified Basics
- Hypernatremia
- Definitions and Risk Factors
- Pathophysiology of Hypernatremia
- Hyponatremia
- Definitions and Risk Factors
- Pathophysiology of Hyponatremia
- Pseudohyponatremia
- Hypovolemic Hyponatremia
- Euvolemic Hyponatremia
- Hypervolemic Hyponatremia
- Differential Diagnosis
- Prehospital Care
- Emergency Department Evaluation
- Hypernatremia History
- Hyponatremia History
- Diagnostic Studies
- Laboratory Studies
- Laboratory Studies for Suspected Hypernatremia
- Laboratory Studies for Suspected Hyponatremia
- Imaging Studies
- Treatment
- Hypernatremia Treatment
- Hypernatremia Treatment Summary
- Hyponatremia Treatment
- Symptomatic Hyponatremia
- Chronic Hyponatremia
- Asymptomatic Hyponatremia
- Hyponatremia in Hypovolemic Patients
- Hyponatremia in Hypervolemic Patients
- Hyponatremia From SIADH
- Overcorrection of Hyponatremia
- Special Populations
- Pediatric Patients
- Pediatric Hypernatremia
- Pediatric Hyponatremia
- Exercise-Associated Hyponatremia
- Seasonal Variations of Hyponatremia
- Controversies and Cutting Edge
- Management of Hyponatremia
- Challenges With Intravenous Fluid Shortages
- Disposition
- 5 Things That Will Change Your Practice
- Risk Management Pitfalls for Sodium Emergencies in the Emergency Department
- Summary
- Time- and Cost-Effective Strategies
- Case Conclusions
- Clinical Pathways
- Clinical Pathway for Management of Hyponatremia in the Emergency Department
- Clinical Pathway for Management of Hypernatremia in the Emergency Department
- Tables and Figures
- References
Abstract
Manifestation of sodium disorders will depend on the rapidity of development, the absolute level of sodium, and the patient’s overall health. Acute symptomatic hypernatremia should be corrected rapidly, while chronic hypernatremia is generally corrected more slowly due to the risks for brain edema during treatment. Hyponatremia symptoms are vague, but acute severe symptomatic hyponatremia, whether self-induced, drug-related, or hospital-acquired, is a medical emergency that demands immediate recognition and intervention. This issue reviews the evidence on the causes, diagnosis, and treatment of hypernatremia and hyponatremia in the emergency department, focusing on early recognition and best-practice management.
Case Presentations
- The patient has had diarrhea and vomiting for the previous 4 days. Current medications include metformin, hydrochlorothiazide, lisinopril, and aspirin.
- His vital signs on arrival in the ED are: oral temperature, 38.7°C; heart rate, 114 beats/min; blood pressure, 86/53 mm Hg; respiratory rate, 24 breaths/min; and oxygen saturation, 94% on 3 L nasal cannula.
- On physical examination, he is cachectic, with dry oral mucous membranes, and is reportedly more confused than his baseline. His abdomen is soft and nontender, with no rebound or guarding. However, he does have an episode of nonbloody, nonbilious emesis during your examination.
- Blood is obtained, and a serum chemistry panel shows: sodium, 154 mEq/L; potassium, 3.9 mEq/L; chloride, 108 mEq/L; bicarbonate, 14 mEq/L; BUN, 55 mg/dL; creatinine, 2.0 mg/dL; and glucose, 112 mg/dL. The nurse asks you what IV fluids you want and how fast...
- Her roommate said she called the ambulance because her friend was vomiting and confused this morning after a long night of drinking alcohol and taking ecstasy. EMS noted multiple empty beer cans and empty shot glasses scattered around the dorm room, but no empty pill bottles were seen.
- On arrival at the ED, the team administered 2 mg IV lorazepam for continued tonic-clonic seizure activity. Her vital signs are: oral temperature, 37.7°C; heart rate, 129 beats/min; blood pressure, 146/64 mm Hg; respiratory rate, 13 breaths/min; and oxygen saturation, 93% on 100% oxygen via nonrebreather mask. Her finger-stick glucose result is 89 mg/dL.
- The patient’s seizures persist, despite a total of 10 mg of lorazepam and a loading dose of levetiracetam administered. The patient is intubated, and shortly thereafter her blood chemistries return, revealing: sodium, 104 mEq/L; potassium, 3.0 mEq/L; chloride, 112 mEq/L; bicarbonate, 16 mEq/L; BUN, 51 mg/dL; creatinine, 1.5 mg/dL; and glucose, 89 mg/dL.
- You realize that the patient’s hyponatremia needs to be emergently corrected, but you wonder: how fast is too fast...
- The patient is protecting his airway and has a bounding radial pulse, but he is confused, has edema of the fingers and wrists, and is vomiting. He has normal skin turgor and color. His running partner states that he had his friend stop at every water stop during the race to make sure he remained hydrated.
- The medic establishes IV access. The patient‘s vital signs include: temperature, 39.0°C; pulse, 138 beats/min; blood pressure, 99/53 mm Hg; and oxygen saturation, 100% via a nonrebreather mask.
- The paramedic you are working with suspects dehydration and tells you that he plans to aggressively rehydrate the athlete with 2 L of normal saline. You have a different plan…
How would you manage these patients? Subscribe for evidence-based best practices and to discover the outcomes.
Clinical Pathway for Managing Patients Presenting with Acute Diarrhea in Urgent Care
Subscribe to access the complete Clinical Pathway to guide your clinical decision making.
Tables and Figures
Subscribe for full access to all Tables and Figures.
Buy this issue and
CME test to get 4 CME credits.
Key References
Following are the most informative references cited in this paper, as determined by the authors.
1. * Otterness K, Singer AJ, Thode HC, Jr., et al. Hyponatremia and hypernatremia in the emergency department: severity and outcomes. Clin Exp Emerg Med. 2023;10(2):172-180. (Retrospective; 57,427 patients) DOI: 10.15441/ceem.22.380
6. * Lindner G, Pfortmüller CA, Leichtle AB, et al. Age-related variety in electrolyte levels and prevalence of dysnatremias and dyskalemias in patients presenting to the emergency department. Gerontology. 2014;60(5):420-423. (Retrospective; 20,667 patients) DOI: 10.1159/000360134
7. * Arampatzis S, Frauchiger B, Fiedler GM, et al. Characteristics, symptoms, and outcome of severe dysnatremias present on hospital admission. Am J Med. 2012;125(11):1125. (Retrospective case series; 77,847 patients) DOI: 10.1016/j.amjmed.2012.04.041
38. * Spasovski G VR, Allolio B. Clinical practice guideline on diagnosis and treatment of hyponatremia. Intensive Care Med. 2014:40(43):320-331. (Review) DOI: 10.1530/EJE-13-1020
44. * Adrogué HJ TB, Madias NE. Diagnosis and management of hyponatremia: a review. JAMA. 2022:328(3):280-291. (Review) DOI: 10.1001/jama.2022.11176
45. * Lindner G SC, Hadinger M. Hyponatremia in the emergency department. Am J Emerg Med. 2022:60:1-8 (Review) DOI: 10.1016/j.ajem.2022.07.023
50. * Sterns RH CJ, Silver SM, et al. Neurologic sequelae after treatment of severe hyponatremia: a multicenter perspective. J Am Soc Nephrol. 1994:4(8):1522-1530. (Multi-center restrospective; 64 patients) DOI: 10.1681/ASN.V481522
51. * Adrogué HJ, Madias NE. Hyponatremia. N Engl J Med. 2000:342(21):1581-1589. (Review) DOI: 10.1001/jama.2022.11176
59. * Brunner JE, Redmond JM, Haggar AM, et al. Central pontine myelinolysis and pontine lesions after rapid correction of hyponatremia: a prospective magnetic resonance imaging study. Ann Neurol. 1990:27(1):61-66. (Prospective; 13 patients) DOI: 10.1002/ana.410270110
Subscribe to get the full list of 81 references and see how the authors distilled all of the evidence into a concise, clinically relevant, practical resource.
Keywords: hypervolemia, hypovolemia, sodium, electrolyte, insipidus, SIADH, hypovolemic, euvolemic, hypervolemic, pseudohyponatremia, diuretic, saline, exercise, fluid